- Home
- Companies
- Akrotome Imaging, Inc.
- Applications
- Clinicians - FIRE – A Flexible and ...
Clinicians - FIRE – A Flexible and Precise Molecular Probe Platform Expressly Designed for Cancer Surgeons - Medical / Health Care
A critical factor in ensuring complete resection of a solid cancer tumor is the ability to determine where diseased tissue ends and healthy tissue begins. Our research, as well as our clinical collaborators, confirm that there is a pressing unmet clinical need: the ability to rapidly and globally assess the status of marginal tissue adjacent to a tumor while the patient is in the OR.
Fluorescent molecular probes which “light up” cancer tissue offer a potentially exciting solution to this problem. A number of companies are working in this area. In general, their probes target cancer-related proteins and require the systemic administration of pharmacologically-significant amounts of probe hours or days before a surgical procedure.
Cancer is a diverse disease, and optimal clinical approaches, as determined by the surgeon, mean that “one size fits all” solutions, are usually suboptimal. We worked directly with cancer surgeons and surgical oncologists to address this unmet need, developing a probe architecture with unmatched flexibility and performance that eliminated the shortcomings of other molecular probes—Akrotome FIRE™.
Our unique technology advantages include:
- Targeting—FIRE targets only active cathepsins over-expressed in the margins of tumors. Activated FIRE glows brightly and precisely delineates the extent of diseased tissue
- Quenching—FIRE is a quenched probe. It activates only when it encounters active cathepsins. Quenching virtually eliminates background fluorescence that is a common problem with other probes
- Speed—FIRE probes activate in minutes, rather than hours or days, meaning they can be administered when needed in the Operating Room
- Unmatched Flexibility—FIRE probes offer a number of administration methods to meet the needs of an individual procedure as determined by the surgeon: in-vivo topical; in-vivo systemic; and ex-vivo topical. The in-vivo topical method, where probe is applied directly into the surgical cavity, is especially attractive as it uses a very small amount of probe, penetrates tissue to a depth of several millimeters, and has the ability to identify non-vascularized cancer tissue.
- World-Class Imager—Our probes are supported by a small, hand-held, highly sensitive imager which is FDA approved for fluorescent surgery applications in the US and in Europe
Because FIRE probes are designed expressly for surgeons, they are built to integrate seamlessly into the clinical workflow of the Operating Room. Blending outstanding performance and flexibility with affordability, the FIRE platform is a superior solution for cancer surgery applications where the determination of the status of marginal tissue is a critical prognostic indicator.
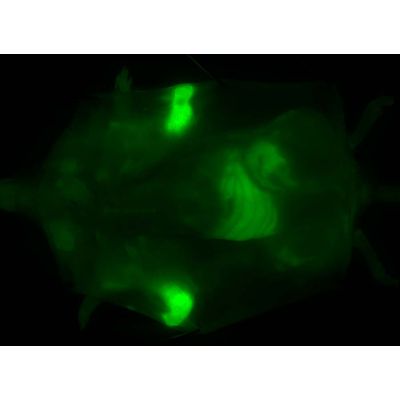
Human breast adenocarcinoma cells were implanted into the chest of an immunocompromised female mouse. When an appropriate size tumor was achieved, the tumor was surgically exposed and FIRE was topically applied to the tumor area (white circles) and adjacent normal tissue (red circle).
Solvent was applied to the other half of tumor tissue (blue circle). After 5 minutes both probe and solvent were removed, tissue was rinsed twice with saline and imaged using the Akrotome handheld camera. Upper right panel preimage indicates no background fluorescence. Lower left panel indicates robustness of signal achieved five minutes after topical application. Living animal under isofloran anesthesia was used for the imaging.

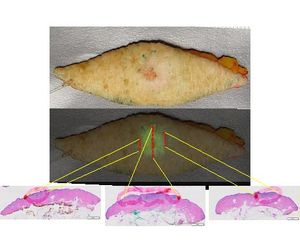
FIRE was topically applied to a resected skin cancer sample. After 10 minutes incubation the imaging device was used to assess probe activation. Directed histological sections were used to correlate cancer (red circles lower panel) to fluorescence signal, overlay data middle panel.
Pathology demonstrates good correlation between the location of fluorescent signal and pathologically confirmed cancer.