Immunoscore for IC in Lung Cancer - Medical / Health Care - Clinical Services
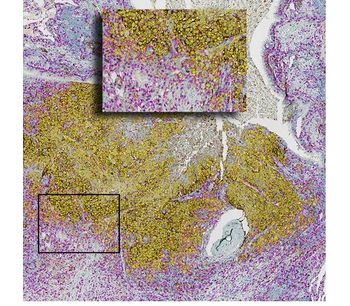
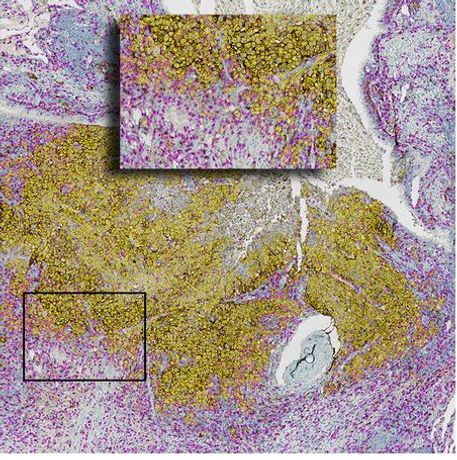
IMMUNOSCORE® IC is intended for the detection of PD-L1 protein and the concomitant detection of CD8 + cells in Non-Small Cell Lung cancer (NSCLC) tissue.
-
Most popular related searches
Details
IMMUNOSCORE® IC, as a member of the IMMUNOSCORE® family assays, is a standardized Immunohistochemistry (IHC)-based assay.
In addition, HalioDx provides image and data analysis services allowing standardized quantification of both PD-L1+ and CD8+ cell populations and the assessment of cells clustering as well as a proximity index. These measures aim to help identifying which NSCLC patients may best respond to anti-PD1 or anti-PD-L1 immunotherapies.
IMMUNOSCORE® IC is available as a CE-IVD assay for PD-L1+ cells detection and as an RUO* solution in the Rest of The World through our comprehensive offer integrating HalioDx service laboratory.
IMMUNOSCORE® IC solution consists in two parts:
- IMMUNOSCORE® IC IHC kit includes ready-to-use anti-PD-L1 and anti-CD8 antibodies and controls for IHC dual-staining. The stained slide is directly readable by the pathologist to determine the percentage of PD-L1 positive tumor cells, evaluate the presence of PDL1+ immune cells and of CD8+ cells.
- Image analysis for complementary descriptive information:
- Standardized computation of CD8+ and PD-L1+ cell densities on the same slide
- PD-L1+ and CD8+ cells clustering
- Proximity index between PD-L1+ and CD8+ cells
IMMUNOSCORE® IC is intended to help clinicians defining immunotherapy treatment strategy for patients with NSCLC.
Anti-PD-1/PD-L1 are now established treatments for advanced NSCLC patients; however, PD-L1 predictive value is not optimal. Association of PD-L1 + TC % with the assessment of tumor-infiltrating lymphocytes (TILs) via CD8+ T cells quantification and proximity measurement should improve the selection of patients likely to respond to Immune Checkpoint Inhibitors treatment.