

Adaptive Designs in nQuery - Case Study
Effective and Efficient Drug Development
Challenge
Increased Cost, Risk, and Competition in the Drug Development Processes
Dr. Luis Rojas, the Executive Director Head of Biostatistics at Target Health, is a subject matter expert in study design and sample size calculations with more than 30 years of industry experience.
He has worked in the leading CROs in the industry and has assisted pharmaceutical companies with the drug development process with clinical trials, programs, and portfolio designs. His experience extends from preclinical, phases I through IV, biosimilar, and bioequivalent using fixed, adaptive designs, stand-alone, and master protocols in multiple therapeutic areas.
Today this work presents new challenges. Drug pipelines are larger than ever before, while the cost of bringing new drugs to market has nearly doubled in the past decade. This trend has compressed drug development ROIs and increased competition.
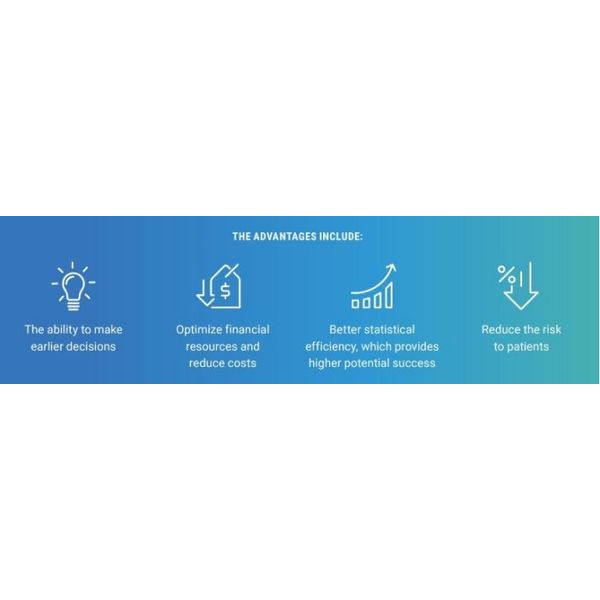
- Increasing trial complexity
- Changing regulations
- Spiraling costs
- Patient access
nQuery is a complete solution for Adaptive Trial Design
With increasing costs, competition, and risk, adaptive clinical trials have made rapid headway as a method to alleviate these challenges. Adaptive trials enable continual modification to the trial design based on interim data. This means that with adaptive trials, you have the opportunity to make changes to your trial, while it is still ongoing. Within nQuery, the world’s most trusted clinical trial design platform, there is a dedicated module for adaptive clinical trials that provide biostatisticians with a range of tools across various adaptive disciplines for sample size calculation.
That’s why Dr. Rojas is a proponent of nQuery to serve as a reliable digital tool for sample size calculations in adaptive and fixed trials.
Leveraging nQuery, Dr. Rojas has developed hundreds of clinical trials and programs.
