

EluNIR DES in a Challenging Case - Case Study
Background:
This paper describes a challenging case of implantation of the EluNIR™ Ridaforolimus eluting elastomeric stent (Medinol Ltd.). This complex case involved a patient with a flow-limiting lesion in a native coronary artery post bypass surgery.
The new EluNIR stent system includes a stent with a low metal footprint that comprises struts of variable width: ultra-narrow struts having a width of 40µm and narrow supportive struts having a width of 72µm. The combination of different strut widths enables the stent to provide both excellent flexibility and conformability, and at the same time adequate radial strength to support the artery after dilation. The stent geometry enables excellent deliverability even in tortuous and calcified lesions. In addition, the delivery system of EluNIR includes an innovative metallic spring tip that was designed to enhance crossability and buckle resistance. Other stent delivery systems that typically have polymer tips, are prone to flare-out and buckle and may impact deliverability.
Case Report:
A 59-year-old man with prior history of coronary bypass surgery and angina was treated at Columbus Clinic Center in Milan, Italy. His coronary angiography performed at a referring center showed a patent Left Internal Mammary Artery (LIMA) bypass graft anastomosed to the mid portion of the native LAD. Distal to the anastomosis (red arrow), there was a long, calcified and tortuous lesion in the native LAD (Figure 1, black arrows). The proximal LAD had a 75% calcified lesion proximal to the LIMA anastomosis. The LAD provided collateral flow to the native RCA that had an occluded venous graft. An attempt to perform PCI at the referring center failed due to inability to dilate the calcified lesion and the patient was subsequently referred for PCI at Columbus Clinic Center.
The case was performed via the right femoral artery. A guidewire was directed through the native LAD across the lesions in the mid LAD into the distal LAD. Simultaneous angiography was performed through the LIMA bypass to better visualize the LAD. A 1.25mm Rotablator burr was advanced through the native LAD to prepare the proximal and mid LAD lesions. Predilatation was successfully performed and following that, a 2.5/24mm EluNIR stent (Medinol Ltd.) was advanced and implanted in the mid LAD lesion. Next, a second EluNIR stent, measuring 2.5/24mm, was deployed in the mid LAD, proximal to the first one towards the LIMA anastomosis. Finally, a 2.25/30mm drug eluting balloon was inflated in the mid-distal LAD.
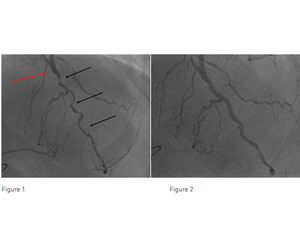
Discussion and Conclusion:
The procedure with the EluNIR stent system (Medinol Ltd.) was successful and the final angiographic result was very good (Figure 2). The EluNIR stent system with the metallic spring tip was highly deliverable; it traversed the tight and severely calcified lesions and managed to navigate the tortuous course of the LAD. Finally, two EluNIR stents were implanted in the native LAD and blood flow was restored.
The new EluNIR stent was recently approved for use in Europe and the United States. The approval in the United States was granted based on the results of the BIONICS trial*, that included 1,919 patients in the United States, Canada, Europe and Israel and compared the EluNIR stent to the Resolute stent (Medtronic). The primary endpoint of target lesion failure at 1 year was 5.4% for EluNIR, which was found non-inferior to Resolute. Definite/probable Stent thrombosis rate at 1 year was very low: 0.4%, with 0% late stent thrombosis beyond 30 days.
