Arterial Disease (Arterial Health) Equipment
-
Manufactured by Orchestra BioMed, Inc.based in USA
Virtue SAB is a patented drug/device combination product for the treatment of artery disease that delivers a proprietary extended release formulation of sirolimus, SirolimusEFR, during balloon angioplasty without the need for a coating or a permanent ...
-
based in USA
PAD is a common circulatory problem in which narrowed arteries reduce blood flow to the extremities – generally affecting one or both legs. It is caused when the inner lining of arteries located outside of the heart get damaged by factors such as smoking, obesity, high blood pressure, elevated cholesterol, elevated triglyceride ...
-
Manufactured by Pharmigene, Inc.based in TAIWAN
Clodpgrel, an antiplatelet agents is indicated for the reduction of thrombotic events as follows: Recent MI, Recent Stroke or Established Peripheral Arterial Disease. For patients with a history of recent myocardial infarction (MI), recent stroke, or established peripheral arterial disease, Clopidogrel has been shown to reduce ...
-
Manufactured by Reflow Medical, Inc.based in USA
Expanded Indication to Cross Chronic Total Occlusions (CTOS) in Peripheral Artery Disease. Compatible with the physician’s choice of guidewire. Works with the Reflow Spex™ and SpexLP™ catheters for added support in lesion ...
-
Manufactured by Atys Medicalbased in FRANCE
SysToe measures toe pressure as part of the diagnosis or monitoring of peripheral arterial disease in the lower ...
-
Manufactured by SOFIEbased in USA
N13-Ammonia is a radioactive diagnostic agent for Positron Emission Tomography (PET) indicated for diagnostic PET imaging of the myocardium under rest or pharmacologic stress conditions to evaluate myocardial perfusion in patients with suspected or existing coronary artery ...
-
Manufactured by SMT Medical Technology GmbHbased in GERMANY
Efficient Testing for Peripheral Arterial Disease (PAD); Arterial ...
-
Manufactured by Reva Medical LLCbased in USA
The MOTIV Bioresorbable Scaffold is designed specifically for use in treating below the knee (BTK) peripheral artery disease. MOTIV is made with Tyrocore™, REVA’s new proprietary polymer. MOTIV is the first bioresorbable scaffold to obtain CE Mark approval for the treatment of BTK disease and has also received FDA ...
-
Manufactured by Atys Medicalbased in FRANCE
Atys has been manufacturing devices for the diagnosis of peripheral vascular disease (peripheral arterial disease (PAD) and venous insufficiency) for 30 ...
-
Manufactured by Concept Medical Inc.based in USA
AN EXCELLENT ALTERNATIVE FOR REVASCULAZATION OF SFA AND BTK ARTERIES.MagicTouch PTA is intended to prevent re-narrowing of superficial femoral and popliteal arteries in patients with peripheral artery ...
-
Manufactured by D. E. Hokanson, Inc.based in USA
The ABI is an easy, noninvasive exam for peripheral arterial disease (PAD) screening. This exam is recommended by the American Diabetes Association, the American Heart Association, the American College of Cardiology and the American College of Physicians as a basic screening examination for PAD. ...
-
Manufactured by Bioprojetbased in FRANCE
Kengrexal, co-administered with acetylsalicylic acid (ASA), is indicated for the reduction of thrombotic cardiovascular events in adult patients with coronary artery disease having undergone a percutaneous coronary intervention (PCI), who have not received an oral P2Y12 receptor inhibitor prior to the intervention and in whom the ...
-
Manufactured by Circle Cardiovascular Imaging Inc.based in CANADA
Specialized tools for the assessment of coronary artery disease using cardiac CT. Read and report Cardiac CT with zero-click coronary artery segmentation and quantifying calcium scoring powered by ...
-
Manufactured by LimFlow, Inc.based in USA
Living with Peripheral Artery Disease (PAD) that progresses to CLTI is challenging. Life revolves around chronic leg pain that can become unbearable over time. The end stage of the disease is characterized by non-healing foot ulcers that considerably lessen mobility and quality of ...
-
Manufactured by Bio Compression Systemsbased in USA
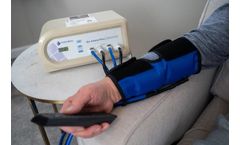
Bio Arterial Plus is an intermittent, high pressure, rapid cycle compression system that sequentially compresses the foot and calf in patients suffering from arterial disease. Through effective, high pressure compression, deep arteries are helped to promote oxygenated blood flow in the legs. ...
-
Manufactured by Concept Medical Inc.based in USA
Novel, First of its kind Sirolimus drug coated balloon catheter for the treatment coronary artery disease. MagicTouch is intended for in-stent restenosis, small vessels, bifurcation lesions and de-novo lesions ...
-
Manufactured by Elixir Medical Corporationbased in USA
The DynamX Drug Eluting Coronary Bioadaptor System is a significant innovation in the treatment of coronary artery disease. Going beyond drug-eluting stents (DES), DynamX represents one of the most significant breakthroughs in implant design in the past 30 ...
-
based in USA
Arterial Disease ...
-
Manufactured by ACME Portable Machines, Inc.based in TAIWAN
ACME Non-invasive Ischemia Detector (ACME NID) is a reliable, non-invasive tool for ischemia detection algorithm. It is a patented algorithm that analyzes 10 second 12 lead ECG captured by either our own QED 2000 or a certified off-the-shelf ECG machine, and it aims to assist in detection of myocardial ischemia related to Coronary Artery ...
-
Manufactured by CARDIONOVUM GmbHbased in GERMANY
Unmatched XLIMUS clinical performance. The ultimate DES for complex artery lesions. XLIMUS is considered as the ultimate coronary DES stent system to treat complex coronary artery disease by reaching and crossing the most challenging ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you