Artery Graft (Arterial Health) Equipment
-
by XELTIS BVbased in NETHERLANDS
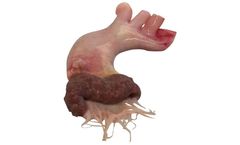
The XABG device is a restorative, synthetic, electrospun blood vessels for CABG surgery, overtime turning into a living vessel made of patient’s own ...
-
Manufactured by XyloCor Therapeuticsbased in USA
Coronary Artery Bypass Graft Surgery or CABG is a procedure used to treat coronary artery disease – the narrowing or blockage of the blood vessels that supply oxygen and nutrients to the heart muscle. During CABG, a healthy artery or vein from the body is connected, or grafted, to the blocked coronary ...
-
Manufactured by UMA Instrumentsbased in USA
The PA-100 is an enhanced Collens Sphygmo-Oscillometer developed to assist physicians in diagnosing peripheral vascular disease and to monitor arterial grafts. UMA Plethysmo Analyzer accurately measures volume changes of blood flow in ...
-
Manufactured by UMA Instrumentsbased in USA
The PA-150 uses state of the art technology, developed to assist the physician in diagnosing peripheral vascular disease, diabetic screening for ischemia, and to monitor arterial grafts. Plethysmography is an indirect measurement of blood flow into a limb. UMA Plethysmo Analyzers accurately measure these volume ...
-
Manufactured by LeMaitre Vascular, Inc.based in USA
Artegraft is a bovine carotid artery graft. Its biological fibrous matrix is processed to enhance long-term patency and provide a tightly woven, cross-linked conduit that is flexible and compliant. For Functional Hemodialysis Access & Lower Extremity ...
-
Manufactured by Celixir plcbased in UNITED KINGDOM
PHASE 2 : Our first investigational cardiac regenerative medicine consists of iMP cells. iMP cells are designed to treat patients with ischaemic heart disease – ischaemic cardiomyopathy – undergoing coronary artery bypass graft (CABG). They are injected around cardiac scar tissue via a single application during bypass ...
-
Manufactured by XyloCor Therapeuticsbased in USA
XC001 is an investigational gene therapy for the treatment of patients with refractory angina with no treatment options. Patients who have exhausted pharmacologic options and are not eligible for PCI or coronary artery bypass grafts become sedentary because of their symptoms and this can exacerbate comorbidities causing rapid ...
-
Manufactured by Carl Zeiss Meditec AGbased in GERMANY
INFRARED 800 from ZEISS enables intraoperative visual assessment of blood flow and vessel patency during arteriovenous malformation (AVM), bypass, aneurysm and anastomosis surgery. ZEISS INFRARED 800 is indicated for use in neurosurgery, plastic and reconstructive procedures and coronary artery bypass graft ...
-
Manufactured by Baymed Inovative Health Products Inc.based in TURKEY
drape layering. Baymed® disposable drapes for cardiovascular surgery offer effective fluid control for a dry working area. Our range includes single drapes and sets developed for cardiothoracic surgery, including heart valve replacements, pacemaker surgery, thoracotomy and coronary artery bypass ...
-
Manufactured by Mölnlycke Health Care ABbased in SWEDEN
All-in-one and split-sheet solutions for a wide variety of procedures. BARRIER® drapes for cardiovascular surgery offer effective fluid control for a dry working area. Our range includes single drapes and sets developed for cardiothoracic surgery, including heart valve replacements, pacemaker surgery, thoracotomy and coronary artery ...
-
based in USA
DuraGraft®- Marizyme’ s flagship product is a first-in-class product used during Coronary Artery Bypass Grafting (CABG surgery) to help prevent vein graft failure and its complications. DuraGraft® is a biocompatible intraoperative vascular graft storage and flushing solution that maintains the ...
-
Manufactured by Revotek Co., Ltd.based in USA
It is estimated by the World Health Organization that the current global prevalence of advanced vascular disease is ~175mm people and increasing. One of the most common clinical interventions to treat advanced vascular disease is the replacement of stenotic or occluded arteries with vascular grafts. This is often accomplished utilizing ...
-
Manufactured by amg International GmbHbased in GERMANY
lesions in coronary arteries and ...
-
Manufactured by LeMaitre Vascular, Inc.based in USA
RestoreFlow Allografts provide a high quality allograft tissue using state-of-the-art technologies for tissue restoration and preparation techniques. These allografts can be used for cardiac repair and reconstruction, as well as for adults with extensive valvular disease. Durable Alternative Valves and ...
-
Manufactured by Mizuho Medical Co., Ltdbased in JAPAN
Over 40 instruments designed for varying depths of cranial bypass ...
-
Manufactured by Merit Medical Systemsbased in USA
Central venous stenosis is a common and serious vascular complication among long-term hemodialysis patients that can lead to the loss of vascular access and a diminished life expectancy. The HeRO Graft (Hemodialysis Reliable Outflow) is the only fully subcutaneous AV access solution clinically proven to maintain long-term access for ...
-
Manufactured by Vascular Flow Technologies Ltdbased in UNITED KINGDOM
Our graft is an expanded polytetrafluoroethylene (ePTFE) graft with removable external beading over its proximal section. The distal portion of the graft contains the patented Spiral Flow™ Inducer and pre-cut distal anastomotic cuff. Any rings on this segment of the graft are NON-REMOVABLE. The Spiral Flow™ Inducer is ...
-
Manufactured by AtriCure, Inc.based in USA
The Isolator Synergy ablation clamp is the FIRST and ONLY surgical ablation device offered with FDA approval for the treatment of Atrial Fibrillation (Afib) and the only device either Catheter or Surgical to be approved for the treatment of persistent and long-standing persistent ...
-
Manufactured by Abena North America, Inc.based in USA
Abena’s Foam Dressing is a hydrophilic polymer with a three-dimensional foam structure that is processed by the latest technological advancements to quickly absorb wound exudate and help maintain a moist wound healing environment. The dressing is designed to not stick to the wound and help minimize user’s discomfort upon removal. ...
-
Manufactured by Carl Zeiss Meditec AGbased in GERMANY
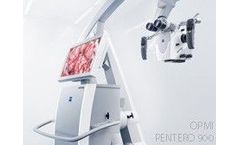
OPMI PENTERO 900 represents the next generation visualization system. Building on the innovations of OPMI Pentero introduced in 2004, OPMI PENTERO 900 combines award-winning design concepts and new functionalities in a proven, fully integrated platform. Key functions have been enhanced and new visualization methods integrated, raising OPMI PENTERO 900 to a high level of ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you