COPD News
-
Showcase
AioCare system could be successfully applied in primary healthcare Centers.
The role of spirometry in diagnosing and monitoring chronic obstructive airway diseases is crucial. According to WHO, the demonstration of irreversible airway obstruction is essential for diagnosing chronic obstructive pulmonary disease (COPD), which is the third leading cause of death worldwide. It is estimated that up to 67–81% of patients with COPD remain undiagnosed. Mobile phone-linked ...
By AioCare
-
Showcase
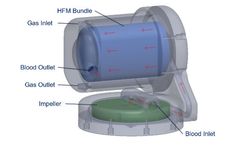
ALung Announces Commercial Development of its Breakthrough Next Generation Artificial Lung
PITTSBURGH–(BUSINESS WIRE)– April 4, 2020 – ALung Technologies, Inc., the leading provider of low-flow extracorporeal carbon dioxide removal (ECCO2R) technologies for treating patients with acute respiratory failure, announced the recent initiation of commercial development of its next generation artificial lung, which expands the Company’s focus on highly efficient gas ...
-
Showcase
New device offers a mouthful of data for chronic diseases
Aidar Health, formerly known as Multisensor Diagnostics, has developed a portable hand-held device that allows patients to measure and track vital signs in a matter of seconds. Roughly the size of an iPhone, MouthLab consists of a hand unit and a mouthpiece that fits between the teeth and lips. The mouthpiece uses special sensors to gather information from breathing patterns, saliva and blood ...
By Aidar Health
-
Vitalograph and MedM Enhance Remote Respiratory Monitoring
Vitalograph has joined forces with MedM Inc. to further enhance patient respiratory monitoring experience with digital health capabilities. The MedM health monitoring app is paired with Vitalograph’s Asthma-1 BT and Lung Monitor BT Smart devices. The app works as a digital asthma or lung function diary, helping users to better understand and manage their condition. Patient diaries like ...
-
Vitalograph introduces tremoflo C-100 oscillometer to its extensive range of respiratory diagnostic solutions
Vitalograph, a global leader in the development and production of respiratory diagnostic solutions and clinical trial services, has launched the Vitalograph tremoflo® C-100 at this year’s ATS International Conference, in Washington. Developed in partnership with Thorasys, the Vitalograph tremoflo® C-100 oscillometer helps to detect the presence of small airway impairment, providing ...
-
Vitalograph introduces tremoflo C-100 oscillometer to its extensive range of respiratory diagnostic solutions
Vitalograph, a global leader in the development and production of respiratory diagnostic solutions and clinical trial services, has launched the Vitalograph tremoflo® C-100 at this year’s ATS International Conference, taking place in Washington. ...
-
Third Pole Therapeutics Receives $32M Strategic Equity Investment to Accelerate its Tankless Inhaled Nitric Oxide (iNO) Platform
Third Pole Therapeutics, a privately held company developing critical life-sustaining cardio-pulmonary therapies, announced today a $32M equity investment, from a large medical device innovator. Bill Athenson, CEO of Third Pole, stated, “We are thrilled to receive this investment, which raises Third Pole Therapeutics’ series B financing and commitments to date to over $85M. I am ...
-
Alex MacKerell, PhD, SilcsBio Co-founder & CSO, Co-Leads Study on Use of SILCS Technology to Improve Lung Disease Treatments
December 14, 2022, Baltimore, MD – SilcsBio, a technology company that develops and markets software and services for structure-based drug design, today announced its Co-Founder and Chief Scientific Officer, Alexander D. MacKerell Jr., PhD, has co-led a study focusing on improving respiratory treatments via SilcsBio’s Site Identification by Ligand Competitive Saturation (SILCS) ...
-
Pulmonx Receives Japanese MHLW Approval of Zephyr Endobronchial Valve for the Treatment of Severe COPD/Emphysema
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, announced today that the Japanese Ministry of Health, Labour and Welfare (MHLW) has approved the Zephyr® Endobronchial Valve for treating severe COPD/emphysema patients following a positive recommendation by Pharmaceuticals and Medical Devices Agency (PMDA). ...
-
Biomark provides business update and second quarter financial results
Vancouver, British Columbia – (November 28, 2022) – BioMark Diagnostics Inc. (“BioMark” or the “Company”) (CSE: BUX) (FSE: 20B) (OTCMKTS: BMKDF) an advanced stage liquid biopsy company with a focus on hard to detect and treat cancers is pleased to reports that it will amend the term of the non-broker warrants (the “Warrants”) issued in relation to ...
-
National COPD Awareness Month in Puerto Rico and Exposure Risks to Respiratory Irritants and Allergens
Chronic obstructive pulmonary disease (COPD), which includes emphysema and chronic bronchitis, makes breathing difficult for millions of Americans who have been diagnosed with the condition. The Centers for Disease Control and Prevention (CDC) reports that millions more suffer from the respiratory disease, but have not been diagnosed or treated. In Puerto Rico and throughout the rest of the ...
-
Pulmonx to Present at the 34th Annual Piper Sandler Healthcare Conference
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, today announced the company will be participating in a fireside chat at the 34th Annual Piper Sandler Healthcare Conference in New York on Wednesday, November 30, 2022, at 11:30 AM PT / 2:30 PM ET. A live and archived webcast of the presentation will be available ...
-
Simeox device is now available in Canada!
We are happy to announce that the #simeox device is now commercially available in Canada. Proud to partner with Novus Medical to help respiratory clinicians improve the life of their patients through access to Simeox. The Simeox device is an efficient and comfortable therapy for distal mucus clearance – for patients suffering from chronic lung conditions associated with chest congestion, ...
By PhysioAssist
-
Beyond Air® Reports Financial Results for the Second Quarter of Fiscal Year 2023
Beyond Air, Inc. (NASDAQ: XAIR) a medical device and biopharmaceutical company focused on developing inhaled nitric oxide (NO) for the treatment of patients with respiratory conditions, including serious lung infections and pulmonary hypertension, and, through its affiliate Beyond Cancer, Ltd., ultra-high concentration nitric oxide (UNO) for the treatment of solid tumors, today announced ...
-
Pulmonx to Participate at Upcoming Investor Conferences
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, today announced the company will be participating in two upcoming investor conferences in New York. Pulmonx management is scheduled to participate in a fireside chat at the Stifel Healthcare Conference on Wednesday, November 16, 2022, at 8:00AM ET. Interested ...
-
VisionGate Focuses on the Importance of Early Detection During Lung Cancer Awareness Month
For many families, November signals the start of the holiday season. It is a special time to gather with loved ones and check-in to make sure everyone is happy and healthy. November is also lung cancer awareness month. VisionGate wants to help you, your family and loved ones learn more about who is at risk, the impact, and the types of lung cancer. Because keeping yourself informed ...
-
VIDA receives 510(k) clearance for deep learning-based enhancements to its LungPrint solution
VIDA Diagnostics, Inc. (“VIDA”), the leader in lung and respiratory intelligence, announced today that it has received 510(k) clearance from the Food and Drug Administration (FDA) for enhancements to its LungPrint solution, including automated deep learning-based lung and lobe segmentation algorithms. This clearance further validates the quality and clinical efficacy of the ...
-
Fluidda and Aptar Pharma Exclusive Collaboration
Fluidda, leader in the field of Functional Respiratory Imaging and developer of the medical device Broncholab®, today announced an exclusive collaboration with Nanopharm, an Aptar Pharma company and a leader in contract research and development services for orally inhaled and nasal drug products (OINDPs). The companies will leverage their respective proprietary technology platforms to help ...
By Fluidda
-
Biomark to present validation data of its multianalyte metabolite panel for lung cancer screening at the esmo congress 2022
Vancouver, British Columbia – (September 8, 2022) – BioMark Diagnostics Inc. (“BioMark”) (CSE: BUX) (FSE: 20B) (OTCMKTS: BMKDF) an advanced stage liquid biopsy company with a focus on hard to detect and treat cancers is pleased to announce today that it’s abstract titled “Metabolomic Profiling for the Early Detection of Lung Cancer” has been ...
-
CONVERT Clinical Trial Data Presented at ERS Demonstrates Early Success of AeriSeal System in Patients with Advanced COPD/Emphysema
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, announces the presentation of interim results from the CONVERT Study at the 2022 European Respiratory Society (ERS) International Conference. Data on the first 40 patients in the study demonstrated that treatment with the AeriSeal System successfully converted the ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you