Extremities News
-
Showcase
VisionQuest Abstract on Diabetic Peripheral Neuropathy Accepted by ADA
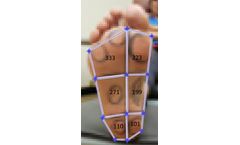
We’re looking forward to presenting “Detecting Diabetic Peripheral Neuropathy Utilizing Thermoregulation of the Plantar Foot” at the upcoming 82nd Scientific Sessions of the American Diabetes Association. Our research aims to detect thermal (infrared) biomarkers of diabetic peripheral neuropathy on the feet of diabetic subjects who have not been diagnosed with DPN. By analyzing ...
-
The Importance of Optimal Medical Therapy (OMT) and Collaborative Care
The HFSA Optimal Medical Therapy in Heart Failure (OMT-HF) program can help your patients achieve their best outcomes. An estimated 6.5 million Americans have heart failure (HF). This year alone another 960,000 will likely be diagnosed with this condition. Chances are several of your patients are in this group. Are they receiving the optimal medical therapy (OMT), that helps them achieve ...
-
Australian Medical Micro Screw Innovation Makes World Debut in 30 Jan 2019 Brisbane Operation
An innovative Australian micro screw created to help significantly reduce complications in the management of orthopaedic injuries and the fixing of fractures in the hand and wrist will be used in a world-first operation in Brisbane by internationally-renowned hand surgeon Dr Greg Couzens on Wednesday 30 January. The Field Micro Screw by Brisbane med-tech start-up Field Orthopaedics has already ...
-
Demand for Brisbane Medical Micro Screw Creates Waitlist Before Official US Launch
The Brisbane medical innovation of the world’s smallest cannulated compression screw by Field Orthopaedics is in high demand in 20 US states before it’s official launch at the American Academy of Orthopaedic Surgeons (AAOS) Annual Meeting from March 12 to 16 in Las Vegas, USA. Hospitals and distributors in 10 US states have requested access to the product before its official launch ...
-
Field Orthopaedics Announces Plans to Debut Award-Winning Micro Screw at AAOS in Las Vegas
Field Orthopaedics, a rapidly growing Australian orthopaedic devices company that focuses on the design and manufacturing of orthopaedic devices to address the shortcomings in the trauma, extremities and biological markets, today announced plans to showcase its FIELD MICRO SCREW SYSTEM at the 2019 American Academy of Orthopaedic Surgeons Annual Meeting in Las Vegas on March 12- ...
-
On the road to IPO, Field Orthopaedics raises $5m for push into US
The rapidly growing Australian orthopaedics devices company, Field Orthopaedics (FO) has just completed a fresh round of fundraising, equipping it with $5million in new capital for a push into the US$11billion trauma and extremities market. As one of the fastest growing market sub-sectors, at a rate of 7-10% per annum, hand and wrist orthopaedics is now around 40% of the upper extremities market. ...
-
OSSIO Receives Vizient Contract for Bio-Integrative Orthopedic Fixation Technology
OSSIO, Inc., an orthopedic fixation technology company, today announced that it has received a contract with Vizient, Inc., making the company’s portfolio of bio-integrative OSSIOfiber products available at negotiated pricing to health care providers in Vizient’s group purchasing organization (GPO) under a three-year contract starting March 1. OSSIO’s contract with Vizient ...
By OSSIO Inc
-
OSSIO Awarded FDA 510(k) Clearance for OSSIOfiber Compression Screw Portfolio
OSSIO, Inc., an orthopedic fixation technology company, today announced that its OSSIOfiber® Compression Screw Portfolio has received 510(k) market clearance from the U.S. Food and Drug Administration (FDA) for maintenance of alignment and fixation of bone fractures, comminuted fractures, fragments, osteotomies, arthrodesis and bone grafts of the upper extremity, fibula, knee, ankle and foot ...
By OSSIO Inc
-
Stanford Surgeon Venita Chandra Joins Alucent Biomedical Scientific Advisory Board as Company Enters Arteriovenous Fistula Market
Alucent Biomedical, a privately held medical technology company founded to transform the way vascular disease is treated, today announced Venita Chandra, M.D., will join the company’s scientific advisory board. Dr. Chandra is board certified in both general and vascular surgery. She is a clinical associate professor of surgery in the Division of Vascular Surgery at Stanford University ...
-
Field Orthopaedics announces strategic alliance with Medartis Inc
The strategic alliance will see the NX Nail system from Field Orthopaedics added to Medartis’ comprehensive hand portfolio in an exclusive distribution partnership, which will extend the global distribution footprint of the product and provide growth opportunities in international markets. With an anatomically inspired head, narrow waist and cutting efficiency, the NX Nail is engineered to ...
-
RevBio Receives a $2 Million NIH Grant to Pursue the Treatment of Wrist Fractures with Its Innovative Bone Adhesive Technology
RevBio, Inc., announced that it has been awarded a Phase II Small Business Innovation Research (SBIR) grant expected to total $2 million over two years from the National Institute on Aging, part of the National Institutes of Health. This funding will allow the company to pursue the treatment of wrist fractures with its patented bone adhesive technology known as Tetranite®. Because the bone ...
By RevBio Inc.
-
Checkpoint Surgical Announces Organizational Changes to Support the Company’s Rapid Growth in the Nerve Care Market Space
Checkpoint Surgical today announced the transition of Len Cosentino from President and CEO to the role of Founder and Executive Advisor. Additionally, several other organizational changes to support the company’s rapid growth have recently been made. Checkpoint Surgical makes surgical devices for intraoperative nerve care. Len Cosentino, who has served as President and CEO from Checkpoint ...
-
The next evolution in blood vessel occlusion
The good news for his patient was that she was young. A lower extremity bypass procedure - like any surgery - can be more difficult as we age. The bad news was that the best veins for the procedure - which involves grafting a vein from another part of the body to bypass her blocked arteries in the leg - were unusable. The patient had undergone varicose vein treatments as a young woman that ...
-
Acumed Acquires Exsomed to Enhance Its Portfolio of Upper Extremity Solutions
Acumed, a Colson Medical - Marmon - Berkshire Hathaway company, is excited to announce the acquisition of ExsoMed, a provider of innovative solutions for hand surgery. The addition of ExsoMed, whose products support an intramedullary approach to treating hand fractures, enhances Acumed’s comprehensive portfolio of upper extremity solutions for simple to complex injuries. Acumed, a Colson ...
-
Rivaroxaban (Xarelto) approved in Japan for patients with peripheral artery disease (PAD) after revascularisation
The Ministry of Health, Labor and Welfare in Japan has approved the use of the oral Factor Xa inhibitor rivaroxaban (Xarelto™) (2.5 mg twice daily, used in combination with aspirin 81-100 mg once daily) to treat patients with peripheral artery disease (PAD) after revascularization. The approval is based on data from the Phase III VOYAGER PAD trial. This study demonstrated a positive ...
By Bayer AG
-
Microbion Corporation Receives up to $2.1 million in Funding Support from the US Navy in Partnership with CUBRC, Inc. to Advance Topical Pravibismane
Microbion Corporation of Bozeman, MT, today announced that it has received non-dilutive funding through its strategic partnership with CUBRC, Inc., a Buffalo-based, independent not-for-profit research company, of up to $2.1 million from the US Navy through the Medical Technology Enterprise Consortium (MTEC) partnership. The funding project is titled “Pravibismane Suspension as a Topical, ...
-
Bluegrass Vascular Announces Featured Paper in the Journal of Vascular Surgery Reporting on the Use of the Surfacer System
Bluegrass Vascular Technologies, a private medical technology company focused on innovating lifesaving devices and methods for vascular access procedures, announced today the publication of a paper featured on the cover of the June 2022 issue of the Journal of Vascular Surgery (JVS) which reports on the use the Surfacer System to perform an Inside-Out® procedure to obtain central venous ...
-
Catalyst OrthoScience Announces Todd Wilson, PhD as Vice President of Medical Education and Training
NAPLES, Fla. - Catalyst OrthoScience, a medical device company focused on the upper extremity orthopedics market, is pleased to announce that Dr. Todd Wilson has joined Catalyst as Vice President of Medical Education and Training effective immediately. “We are excited to have an executive with Todd’s experience join our team at Catalyst OrthoScience,” said Catalyst ...
-
CVRx Receives MR-Conditional Labeling Approval for its Barostim Heart Failure System
MINNEAPOLIS, MAY 9,2022 - CVRx, Inc. (NASDAQ: CVRX) ("CVRx"), developer of the world's first FDA-approved neuromodulation device to treat the symptoms of heart failure, has received U.S. Food and Drug Administration (FDA) approval for magnetic resonance (MR) conditional labeling for its Barostim System. The Barostim System now includes instructions to allow for safe MRI scans of the head and ...
By CVRx
-
Alucent Biomedical Announces First Patient Enrolled in First in Human Natural Vascular Scaffolding Clinical Trial
Alucent Biomedical Inc. announced that it has enrolled the first patient in ACTIVATE II, an Australia-based First-in-Human clinical trial to evaluate the safety and efficacy of its revolutionary Natural Vascular Scaffolding (AlucentNVS) technology. The therapy is designed to open vessels and maintain patency without the use of permanent implants for the treatment of peripheral artery disease ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you