Haemorrhage News
-
Showcase
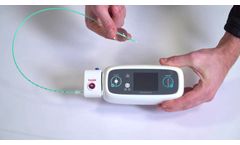
Neurescue`s Breakthrough Intelligent Balloon Catheter FDA 510(k) Cleared for Hemorrhage Control and IDE Approved for Cardiac Arrest
COPENHAGEN, Denmark--(BUSINESS WIRE)--Neurescue, a medical device company developing innovative cardiovascular solutions to improve the outcomes for emergency patients, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to market the company’s NEURESCUE® device for temporary occlusion of large vessels, including patients requiring emergency ...
By Neurescue
-
An Easy, Predictable, and Simpler Way to Embolize Tumors
Instylla is now enrolling patients in a clinical trial for the world’s first hydrogel-based liquid embolic intended for the treatment of hypervascular tumors. A second initial application for its Embrace embolic therapy is hemorrhage control, where the product offers advantages in terms of simplicity, speed, and safety. Amar Sawhney and his teams, it seems, can make medical hydrogels do ...
-
Biotricity wins NIH Grant from the National Heart, Blood, and Lung Institute for AI-Enabled real-time monitoring and predictive analytics for stroke due to chronic kidney failure
Biotricity Inc. (NASDAQ:BTCY) (“Biotricity” or the “Company”), a medical diagnostic and consumer healthcare technology company, today announced that it will be carrying out an NIH-sponsored Phase I study of Bioflux-AI, Biotricity’s innovative artificial intelligence-driven (‘AI’) system of real-time monitoring and prediction technology, in subjects with ...
-
Amsel Medical Announces $1.5 million AFWERX TACFI Grant Award
Amsel Medical Corporation today announced receipt of award and signing of a contract for a non-dilutive AFWERX Tactical Funding Increase (TACFI) of $1.5 million augmenting its original $1M Phase II SBIR Grant. This effort is sponsored by the Air Force Research Laboratory and will enable Amsel Medical to bridge its Phase II efforts towards Phase III commercial scaling. It facilitates the delivery ...
-
Rapid Medical Gains FDA Clearance for the Smallest & Only Adjustable Thrombectomy Device
Rapid Medical, a leading developer of advanced neurovascular devices, announces FDA 510(k) clearance for TIGERTRIEVER 13 for large vessel occlusions at the 2022 Society of NeuroInterventional Surgery’s (SNIS) 19th Annual Meeting in Toronto. TIGERTRIEVER 13 is the smallest revascularization device in the world to date and is designed to remove thrombus from delicate brain blood vessels ...
-
Rapid Medical Marks First Procedures with Numen Coil from MicroPort Scientific
Rapid Medical, a leading developer of advanced neurovascular devices, announces the expansion of its portfolio in the United States and the first Numen™ coil embolization procedure. Rapid Medical now has an exclusive distribution agreement with MicroPort NeuroTech, a subsidiary of MicroPort Scientific Corporation. MircoPort’s global presence offers nearly 300 medical solutions to ...
-
Rapid Medical Enrolls the First Patient in the DISTALS Stroke Trial–Offering Treatment to New Ischemic Stroke Patient Population
Rapid Medical, a leading developer of advanced neurovascular devices, announced today the enrollment of the first patient in the DISTALS Study. The trial is the first-ever FDA investigational device exemption (IDE) trial to examine the safety and effectiveness of mechanical thrombectomy in distal stroke. “Enrolling the first patient in this landmark study is an important milestone for ...
-
Sydney MedTech Baymatob completes capital raise to advance life-saving labour monitoring device
Sydney medical device company Baymatob has completed a placement of $4.215 million, with proceeds used to continue in-human clinical trials for its AI-guided labour monitoring device, Oli. Cornerstoned by Australian Unity’s Future of Healthcare Fund, the placement was strongly supported by Australian institutional and high net worth investors, securing long-term capital partners to support ...
-
Aidoc Showcases Full Ai Care Coordination Suite At International Stroke Conference 2022
AI-powered neuroscience solutions for intracranial hemorrhage, ischemic stroke, cervical spine fractures, on display at ISC 2022, New Orleans, February 9-11, Booth #801 New York, February 8, 2022 – Aidoc, the leading provider of healthcare AI solutions, will be delivering a live demonstration of its comprehensive AI care coordination suite at ISC 2022, February 9-11, in New Orleans, Booth ...
By Aidoc
-
Aidoc Partners With Novant Health, Providing Imaging Ai To Expedite Treatment For Patients In The Emergency Department
Novant Health’s integration of Aidoc’s AI solutions, amid the latest wave of the Omicron variant, expands upon its existing slate of innovative technologies designed to improve delivery of patient care and outcomes Aidoc, the leading provider of enterprise-grade AI solutions for medical imaging, announces a partnership with Novant Health, a health network of over 1,800 physicians ...
By Aidoc
-
JenaValve Announces Strategic Investment and Licensing Agreement with Peijia Medical Limited
JenaValve Technology, Inc. (“JenaValve” or the “Company”), developer and manufacturer of differentiated transcatheter aortic valve replacement (TAVR) systems, today announced a strategic investment and exclusive technology licensing agreement with Peijia Medical Limited (HKEX: 9996) in China. Under the terms of the agreement, Peijia has made a cash and equity investment in ...
-
New Research Demonstrates Clinical Utility of Daxor Corporation Blood Volume (BVA-100) Diagnostic in Neurocritical Care
Data Highlight Daxor’s Diagnostic Changes Care in 69% of Patients With Non-Traumatic Subarachnoid Hemorrhage (nSAH) Daxor Corporation (NYSE MKT: DXR), the global leader in blood volume measurement technology, today announces new data demonstrating the clinical utility of the BVA-100 blood test in patients with non-traumatic subarachnoid hemorrhage (nSAH) presented at the virtual ...
-
Aidoc And Screenpoint Medical Announce Partnership To Provide Complete Ai Solution For Breast Imaging
Aidoc’s comprehensive AI continues to expand to cover the majority of radiology adult subspecialties Aidoc, the leading provider of AI for medical imaging, and ScreenPoint Medical, leader of deep learning AI for 2D and 3D mammography, today announced a collaboration which will incorporate ScreenPoint’s capabilities into Aidoc’s platform. Breast imaging specialists will now be ...
By Aidoc
-
Aidoc And Riverain Technologies Partner To Streamline Access To Time Saving Chest Ai Solutions
Partnership provides a seamless workflow to improve patient outcomes while enabling radiologists to read faster with commercially available solutions. Aidoc and Riverain Technologies, two leading providers of AI for medical imaging, announced a new collaboration today. This new partnership will improve provider access to both solutions by incorporating Riverain’s commercially available ...
By Aidoc
-
Sense receives clearance to expand ongoing clinical trial to evaluate non-invasive brain scanner to sites in India and Canada
Clinical trial underway at five US hospital sites to evaluate Sense’s novel, non-invasive brain scanner to monitor intracranial hemorrhage in acute hospital setting Drug Controller General of India (DCGI) and Health Canada grant permission to conduct clinical trial at multiple sites in India and Canada Current standard of care lacks a means to monitor brain injury continuously, ...
-
Neurolutions IpsiHand Stroke Rehabilitation Device Authorized by FDA
Today, the U.S. Food and Drug Administration authorized marketing of a new device indicated for use in patients 18 and older undergoing stroke rehabilitation to facilitate muscle re-education and for maintaining or increasing range of motion. The Neurolutions IpsiHand Upper Extremity Rehabilitation System (IpsiHand System) is a Brain-Computer-Interface (BCI) device that assists in rehabilitation ...
By Neurolutions
-
Imperative Care Appoints Chief Operating Officer Daniel Davis as President and Member of Board of Directors
Imperative Care, Inc., a company singularly dedicated to addressing unsolved problems in stroke, today announced that its Chief Operating Officer, Daniel Davis, has been appointed as the company’s President. Mr. Davis will oversee the growth of the company’s technology portfolio for the treatment of ischemic and hemorrhagic stroke. He will retain his role as COO and join the ...
-
Instylla Announces Completion of Series B Financing
Instylla, Inc., a privately held company developing minimally invasive liquid embolic products designed for use in peripheral vascular embolotherapy today announced closure of $25.4 million Series B financing. Participants in this latest round of financing included the new investors Excelestar Ventures, Catalyst Health Ventures, and Amed Ventures. Existing investors Ascension Ventures, Sparta ...
-
Cerus Endovascular Receives CE Mark Approval for its Contour 021 device
Cerus Endovascular Ltd., a privately-held, commercial stage medical device company, today announced that it has received CE Mark approval for its state of the art CerusEndo MC 021 microcatheter, designed to allow physicians to access tortuous neurovasculature and deliver therapeutic devices to intended targets. “We remain committed to meeting the ever-increasing needs of the interventional ...
-
Imperative Care Announces $85 Million Series C Financing for Innovative Stroke Treatments
Imperative Care, Inc. today announced that it has completed the initial closing of a Series C financing of $85 million to support the commercial launch of the company’s portfolio, which is composed of the latest advances in neurovascular devices treating ischemic and hemorrhagic strokes. The round was co-led by new investors Ally Bridge Group and Bain Capital Life Sciences. Existing ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you