Intensive Care Unit (ICU) News
-
Showcase
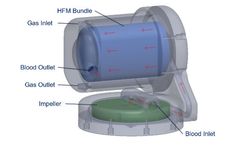
ALung Announces Commercial Development of its Breakthrough Next Generation Artificial Lung
PITTSBURGH–(BUSINESS WIRE)– April 4, 2020 – ALung Technologies, Inc., the leading provider of low-flow extracorporeal carbon dioxide removal (ECCO2R) technologies for treating patients with acute respiratory failure, announced the recent initiation of commercial development of its next generation artificial lung, which expands the Company’s focus on highly efficient gas ...
-
Infection Control Resources to Safeguard Patients and Building Occupants from Acinetobacter and Other Pathogens
The Centers for Disease Control and Prevention describe Acinetobacter as a group of bacteria commonly found in the environment in places like soil and water. While there are many types, the most frequent cause of infections in humans is known as Acinetobacter baumannii. Those most at risk of infection according to the CDC include patients in hospitals, especially those who: · are ...
-
Practicing in the Digital Age: The Benefits of the SurgiCam HD
Summary The SurgiCam® HD, a specialized point-of-view camera designed for clinicians, plays a pivotal role in the documentation, education, and analysis of surgical, dental, and veterinary procedures. Its high-quality image and video capturing capabilities make it an indispensable tool in these fields. This innovative camera significantly enhances surgical practices, education, and research ...
-
With Policy Support, the Market for Surgical Robots Grows
Recently, the Ministry of Industry and Information Technology, the National Health Commission and other 17 departments jointly issued the "robot" application action implementation plan to encourage the development of surgery, auxiliary examination, auxiliary rounds, intensive care, rehabilitation and other medical robot products, "robot The "robot" has ushered in a new round of development ...
-
China to see one longer, more severe COVID wave as Lunar Festival fuels outbreak
Airfinity has updated its cases and deaths forecast for China’s COVID-19 outbreak as the Lunar New Year holiday hastens the spread of the virus. Today’s new forecast considers reports that some provinces including Henan, Gansu, Qinghai and Yunnan have already seen infections peak. Our analytics indicators suggest that the virus has spread more quickly to rural areas, partly driven ...
-
1st Patient treated with Vetex Thrombectomy Catheter
Vetex™ Medical Ltd., an innovator in deep vein thrombosis (DVT) treatment, announced it’s first patient on December 18th, 2018, treated in the VETEX (Venous Thrombus Extraction) Study, a first-in-man, multicenter study of the Vetex™ Thrombectomy Catheter. The Vetex device is the first DVT device to combine rotational and grasping action to quickly and gently remove large volumes ...
-
7 December 2022 - FDA/CDC highlight need for infection preventions
Particular support for decolonisation of pathogens which is the approach taken by its XF-73 Nasal programme Destiny Pharma plc (AIM: DEST), a clinical stage innovative biotechnology company focused on the development of novel medicines that can prevent life-threatening infections, today reports on the publication of findings from the US Food and Drug Administration (FDA) and US Centers for ...
-
BioIntelliSense Acquires AlertWatch Clinical Intelligence Engine to Expand its Comprehensive Portfolio of Continuous Patient Monitoring Solutions
BioIntelliSense, a continuous health monitoring and clinical intelligence company, today announced its acquisition of AlertWatch®, an FDA-cleared, clinically proven patient monitoring solution. AlertWatch uniquely provides robust integration, data analysis and a patented intuitive display of contextual patient data for more efficient, actionable clinical decisions. As part of the recently ...
-
Inari Medical Announces Results from the Fully Enrolled 800-patient US Cohort of the FlowTriever FLASH Registry
Inari Medical, Inc. (NASDAQ: NARI) (“Inari”) a medical device company focused on developing products to treat and transform the lives of patients suffering from venous and other diseases, announced positive outcomes of the fully enrolled 800-patient FLASH registry in pulmonary embolism (“PE”). The data was presented during a Late-Breaking Clinical Trial session at the 2022 ...
-
Positive Clinical Results for Bluejay’s Symphony IL-6 Test Presented at AACC 2022
Demonstrated 98% NPV1 to Identify COVID-19 Patients at Risk for Severe Illness Study Data Indicate Symphony IL-6 Test Could be a Rapid and Precise Diagnostic Platform for Near-Patient Management of Critical Care Bluejay Diagnostics, Inc. (NASDAQ: BJDX) (“Bluejay” or the “Company”), a medical diagnostics company focused on developing cost-effective, rapid, ...
-
N8 Medical Announces First-in-Human Use of its CeraShield™ Endotracheal Tube
N8 Medical, LLC (N8 Medical) today announced its first-in-human clinical trials of its flagship CeraShield™ endotracheal tube (CeraShield™ ETT) for intensive care unit patients expected to require mechanical ventilation for more than 48 hours. N8 Medical is encouraged by preliminary study data and believes that the CeraShield™ ETT will demonstrate safety and a significant ...
-
Biocartis Announces Presentation of Three Idylla™ Studies at 32nd European Congress of Clinical Microbiology & Infectious Diseases (ECCMID)
Biocartis Group NV (the ‘Company’ or ‘Biocartis’), an innovative molecular diagnostics company (Euronext Brussels: BCART), today announces that three Idylla™ study posters, of which two on the SeptiCyte® RAPID (developed in collaboration with Immunexpress) and one on the Idylla™ SARS-CoV-2 Test (CE-IVD) and Idylla™ SARS-CoV-2/Flu/RSV Panel (CE-IVD), ...
By Biocartis NV
-
Micromed Group and Neuroevolution strong together
Micromed Group presents their product lines together with our local distributor at the 34th ENE Congress and the XXIV Congresso Nacional de Medicina Intensiva in Porto.Micromed Group and Neuroevolution in Portugal For the region of Portugal, Micromed Group is working closely with our local distributor Neuroevolution. The team is supporting us on site in distributing the whole portfolio of the ...
-
OpGen Announces Publication of Results from Major Clinical Study Using Unyvero Hospitalized Pneumonia (HPN) Panel in the Lancet Respiratory Medicine
208 patients enrolled in a prospective, randomized, controlled, multicenter, interventional trial Unyvero reduced the use of inappropriate antibiotic therapy by 45%, shortened inappropriate antibiotic therapy by 39 hours, and reduced overall antibiotic therapy duration by 22.5% Unyvero results combined with antibiotic stewardship are efficient and safe in decreasing time on inappropriate ...
By OpGen, Inc.
-
Palisade Bio Announces Inducement Awards Under Inducement Plan For Key New Hires
Palisade Bio (Nasdaq: PALI), a clinical-stage biopharmaceutical company advancing therapies for acute and chronic gastrointestinal complications, today announced that it had made equity grants to four new employees under its 2021 Inducement Plan. On May 4, 2022, the Company’s Compensation Committee granted options to four new non-executive employees covering an aggregate of 140,000 shares ...
By Palisade Bio
-
The Neotec Program finances a new transport ventilator with ICU benefits
INBENTUS , with name Iconic Solutions by Murcia SL, is carrying out a project co-financed with European funds from the Recovery and Resilience Mechanism and "Subsidized by the CDTI" supported by the Ministry of Science and Innovation within the NEOTEC PROGRAM , called " TRANSPORT RESPIRATOR WITH ICU BENEFITS ” SNEO-20211086 The project runs from January 1, 2022 to December 31, ...
By Inbentus
-
Palisade Bio Announces $2.0 Million Registered Direct Offering
Palisade Bio, Inc. (Nasdaq: PALI) (the “Company” or “Palisade Bio”), a clinical-stage biopharmaceutical company advancing therapies for acute and chronic gastrointestinal complications, today announced that it has entered into definitive agreements with several institutional and accredited investors for the purchase and sale in a registered direct offering of 3,646,690 ...
By Palisade Bio
-
Palisade Bio And Newsoara Receive NMPA Clearance To Commence Phase 3 Clinical Trial In China Evaluating LB1148 To Accelerate The Return Of Bowel Function Following Abdominal Surgery
NMPA clearance expands global Phase 3 clinical development program; Company recently announced clearance from U.S. Food and Drug Administration to commence Phase 3 study Newsoara to fully fund Phase 3 program in China Under the Co-Development Agreement, commencement of Phase 3 program in China will trigger milestone payment to Palisade Bio Palisade Bio (Nasdaq: PALI), a clinical stage ...
By Palisade Bio
-
Palisade Bio (Nasdaq: PALI) To Participate In The Virtual Investor Management And KOL Roundtable Event
- Palisade Bio (Nasdaq: PALI), a clinical stage biopharmaceutical company advancing therapies for acute and chronic gastrointestinal (GI) complications, today announced that members of management will participate in the Virtual Investor Management and KOL Roundtable Event on Tuesday, May 3, 2022 at 10:00 AM ET. Participating from the Palisade Bio management team will be Tom Hallam, Ph.D., Chief ...
By Palisade Bio
-
CytoSorbents to Present at the Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum and Jefferies London Healthcare Conference
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces its participation in two upcoming investor conferences. CytoSorbents will present at the Canaccord Genuity Virtual MedTech, Diagnostics and Digital Health & Services Forum on Thursday, November 18, 2021. Dr. ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you