Limb Disorders News
-
Showcase
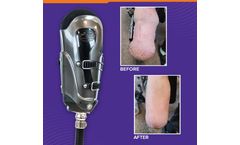
The iFIT device gently and comfortably reshapes the residual limb.
This patient reduced their volume within a month period of time using the iFIT transtibial prosthesis. The iFIT system relieves swelling and shapes the limb quickly. There is no need to wait for prosthetic shrinker socks to reduce volume, so those with limb loss can be fit right away and get back on their feet. ...
-
Showcase
IFIT Helps Persons with Limb Loss in Botswana
This summer, Dr. Timothy Dillingham and his son Rhys visited Botswana with a goal to help persons with limb loss obtain a comfortable, durable socket. Through the UPenn-Botswana Partnership they visited several locations and fit 17 persons in a 7 day time period. They had four hockey bags loaded with these modular devices and a backpack with hand tools. Many of these persons were not able to ...
-
Amsel Medical Announces $1.5 million AFWERX TACFI Grant Award
Amsel Medical Corporation today announced receipt of award and signing of a contract for a non-dilutive AFWERX Tactical Funding Increase (TACFI) of $1.5 million augmenting its original $1M Phase II SBIR Grant. This effort is sponsored by the Air Force Research Laboratory and will enable Amsel Medical to bridge its Phase II efforts towards Phase III commercial scaling. It facilitates the delivery ...
-
Reflow Medical Completes DEEPER LIMUS Clinical Trial Enrollment
Reflow Medical, Inc., a California-based medical device company, has completed enrollment in the DEEPER LIMUS clinical trial (NCT04162418) to evaluate the Temporary Spur Stent System, a patented device designed to treat long, diffuse and severely calcified infrapopliteal disease. The system allows for uniform expansion of the stent to maximize lumen diameter and may reduce acute vessel recoil ...
-
Rivaroxaban (Xarelto) approved in Japan for patients with peripheral artery disease (PAD) after revascularisation
The Ministry of Health, Labor and Welfare in Japan has approved the use of the oral Factor Xa inhibitor rivaroxaban (Xarelto™) (2.5 mg twice daily, used in combination with aspirin 81-100 mg once daily) to treat patients with peripheral artery disease (PAD) after revascularization. The approval is based on data from the Phase III VOYAGER PAD trial. This study demonstrated a positive ...
By Bayer AG
-
Reva Medical Presents 6-Month Results from Motiv BTK Pilot Study
REVA Medical, LLC. (“REVA” or the “Company”), a leader in bioresorbable polymer technologies for vascular applications, is pleased to announce the presentation of the 6- month clinical outcomes data from a post market study of the Company’s MOTIV Below The Knee (BTK) scaffold. Prof. Thomas Rand, MD from the Klinik Floridsdorf Vienna, Austria presented the initial ...
-
Hemostemix Announces Pete Pavlin, P.Eng., Vice-President Operations
Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM; OTC: HMTXF; FSE: 2VFO.F) is pleased to announce the appointment of Mr. Peter Pavlin, P.Eng. to the position of Vice President Operations. Mr. Pavlin earned his Bachelor of Applied Science and Master of Engineering degrees in Mechanical Engineering from the University of Toronto, Canada’s top ranked ...
-
Enrollment Completed In Reflow Medical’s DEEPER OUS Clinical Trial
Reflow Medical, Inc. announces that it has completed patient enrollment in the DEEPER OUS clinical trial (NCT03807531) for the company’s Temporary Spur Stent System, a novel device that features a retrievable stent designed for complex infrapopliteal disease. 106 patients have now been enrolled in the prospective, nonrandomized trial in multiple centers in Europe and New Zealand. Intended ...
-
Capricor Therapeutics Reports First Quarter 2022 Financial Results and Provides Corporate Update
Executed Partnership with Nippon Shinyaku for the Commercialization and Distribution of CAP-1002 for Duchenne Muscular Dystrophy in the United States Upfront Payment of $30 Million Received in 1 st Quarter Strengthens Cash Position and Extends Cash Runway Positive Phase 2 Results Published in The Lancet Evaluating CAP-1002 in Late-Stage Duchenne Muscular Dystrophy Pivotal Phase 3 Trial ...
-
Hemostemix Announces Rick Groome as Special Advisor to the CEO
Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM; OTC: HMTXF; FSE: 2VFO.F) is pleased to announce Mr. Richard (“Rick”) Groome has joined Hemostemix as a special advisor to the CEO, focused on completing a series of financing transactions. Over the last three decades Mr. Groome was instrumental in creating and building two securities firms, Marleau ...
-
Blogpost – The arrival of REAtouch Lite at Axinesis
Axinesis expands its intensive functional rehabilitation offer We are delighted to announce the launch of our new REAtouch® Lite: the device for decentralized upper-limb rehabilitation in the rehabilitation centre or at home. REAtouch Lite is the portable version of the REAtouch: our device for intensive bimanual functional upper-limb rehabilitation. REAtouch bimanual intensive therapy for ...
By Axinesis
-
Blogpost - Scientific proof of the effectiveness of the REAplan
Robot-assisted therapy (RAT) is of significant interest in early rehabilitation, when neuroplasticity is high but motor control is frequently insufficient for patients to independently practice functional movements. Many patients with stroke experience persistent upper limb (UL) impairments. This single-blind, randomized, controlled trial[i] explored the impact of partially substituting ...
By Axinesis
-
Hemostemix Announces Settlement Agreement
Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM; OTC: HMTXF; FSE: 2VFO.F) Calgary, Alberta — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM; OTC: HMTXF; FSE: 2VFO.F) is pleased to announce that it has entered into a settlement agreement (the “Settlement Agreement”) with Aspire Health Science, LLC ...
-
National Institute for Health and Care Excellence (NICE) Publishes Briefing on ReStore Soft Exo-Suit in the UK
ReWalk Robotics, Ltd. (Nasdaq: RWLK) ("ReWalk" or the "Company"), a manufacturer of robotic medical devices for individuals with lower limb disabilities, today announced its ReStore Soft Exo-Suit was the subject of a recent Medtech Innovation Briefing (MIB) by the UK's National Institute for Health and Care Excellence (NICE). These briefings are designed to support National Health Services (NHS) ...
-
The Lancet Publishes Positive Results from Capricor Therapeutics` Phase 2 Study Evaluating CAP-1002 in Late-Stage Duchenne Muscular Dystrophy
One-Year Final Data Highlights Lead Cell Therapy Asset, CAP-1002, for Safety and Efficacy in Slowing Upper Limb and Cardiac Function Deterioration in Patients Data Sets Stage for Pivotal Phase 3 HOPE-3 Clinical Trial Capricor Therapeutics (NASDAQ: CAPR), a biotechnology company focused on the development of transformative cell and exosome-based therapeutics for the treatment and prevention ...
-
Capricor Therapeutics Reports Fourth Quarter and Full Year 2021 Financial Results and Provides Corporate Update
CAP-1002 Cell Therapy Programs Pivotal Phase 3, HOPE-3 Trial Initiation Underway- Entered Exclusive Partnership with Nippon Shinyaku for Commercialization and Distribution of CAP-1002 for Duchenne Muscular Dystrophy in United States- Capricor to Receive Upfront Payment of $30 Million with Additional Milestone Payments of up to $705 Million- Phase 2 INSPIRE Top-Line Data Expected by End of ...
-
Hemostemix Announces Closing of UNIT Private Placement
Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV: HEM; OTC: HMTXF) announces that it has closed its previously announced non-brokered private placement of units (“Units”) announced on February 2, 2022, for gross proceeds of $1,204,849.92 (the “Offering”). The Offering consisted of the issuance of an aggregate of 8,606,071 Units at a price of ...
-
Announcing Key Leadership Team Additions and Scientific Clinical Advisory Board Formation
Advanced Oxygen Therapy Inc. (AOTI), the leader in noninvasive sustained wound healing solutions, announced today a number of significant additions to its Leadership Team, bringing to the company over 100 years of combined strategic experience: Chief Medical Officer – Dr. Matthew G. Garoufalis is Co-Chair of the Alliance of Wound Care Stakeholders and past President of both the American ...
-
BioXtreme and Advanced Medical Solutions s.r.o sign cooperation agreement in Czech Republic and Slovakia
BioXtreme is proud to announce a new cooperation agreement was recently signed with Advanced Medical Solutions s.r.o (AMS) from Brno, Czech Republic. The agreement situates AMS as BioXtreme’s representative in the Czech Republic and Slovakia, with exclusive distribution rights for BioXtreme’s neurorehabilitation products and forming connections with hospitals and rehabilitation ...
-
STEPS Budapest Center for Robotic Rehabilitation to Install deXtreme 3.1 for Commercial Evaluation
BioXtreme is proud to announce that a new commercial evaluation agreement was signed with STEPS Budapest Center for Robotic Rehabilitation. The agreement includes a 6- months evaluation period for deXtreme, by the end of which STEPS commits to purchasing the device (pending evaluation criteria are met). BioXtreme has developed a ground-breaking robotic device for motor-learning, based on ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you