Respiratory Failures News
-
Showcase
ALung Announces Commercial Development of its Breakthrough Next Generation Artificial Lung
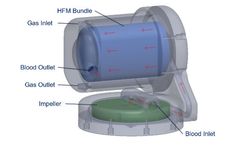
PITTSBURGH–(BUSINESS WIRE)– April 4, 2020 – ALung Technologies, Inc., the leading provider of low-flow extracorporeal carbon dioxide removal (ECCO2R) technologies for treating patients with acute respiratory failure, announced the recent initiation of commercial development of its next generation artificial lung, which expands the Company’s focus on highly efficient gas ...
-
Research on Safe Nebulizer Inhalation Therapy Using Clean Booth and Particle Counters
Particles Plus® instruments have once again been used by scientists for research into Nebulizer Inhalation Therapy. To test the administration of safe Nebulizer Inhalation Therapy, researchers monitored particle concentrations for different particle sizes with Particles Plus® Optical Particle Counters (OPC). Optical Particle Counters can measure particle size distribution and particle ...
-
Aerami Therapeutics to Present at the LifeSci Partners Summer Symposium
Aerami Therapeutics, Inc. (the “Company”) announced today that Anne Whitaker, Chief Executive Officer, will present a corporate overview at the Private Healthcare Company Virtual Summer Symposium, hosted by LifeSci Partners, on August 4 and 5, 2020. Please click the following link to register Presentation Details: Date: August 5, 2020 Time: 9:00 am Eastern Time About Aerami ...
-
China’s COVID-19 wave forecast to have two peaks where cases could reach 4.2 million a day
China is predicted to see two peaks in cases as COVID-19 spreads throughout the country, the first peak in mid-January and the second in early March. New modelling by Airfinity has examined data from China’s regional provinces. The current outbreak is growing more rapidly in some regions than in others. Cases are currently rising much more quickly in Beijing and Guangdong. Using the ...
-
Third Pole Therapeutics Receives $32M Strategic Equity Investment to Accelerate its Tankless Inhaled Nitric Oxide (iNO) Platform
Third Pole Therapeutics, a privately held company developing critical life-sustaining cardio-pulmonary therapies, announced today a $32M equity investment, from a large medical device innovator. Bill Athenson, CEO of Third Pole, stated, “We are thrilled to receive this investment, which raises Third Pole Therapeutics’ series B financing and commitments to date to over $85M. I am ...
-
Eikonoklastes Therapeutics and Forge Biologics Announce Viral Vector Contract Development and AAV Manufacturing Partnership
Eikonoklastes Therapeutics, a preclinical biotech company developing treatments for today’s most challenging diseases, and Forge Biologics, a gene therapy-focused contract development and manufacturing organization, today announced a manufacturing partnership that will advance Eikonoklastes’ AAV-based gene therapy, ET-101, into clinical trials for the treatment of patients with ...
-
Beyond Air® Reports Financial Results for the Second Quarter of Fiscal Year 2023
Beyond Air, Inc. (NASDAQ: XAIR) a medical device and biopharmaceutical company focused on developing inhaled nitric oxide (NO) for the treatment of patients with respiratory conditions, including serious lung infections and pulmonary hypertension, and, through its affiliate Beyond Cancer, Ltd., ultra-high concentration nitric oxide (UNO) for the treatment of solid tumors, today announced ...
-
Honoring Ony’S Founder, Dr. Edmund Egan
Earlier this year, our founder and longtime Chief Medical Officer, Dr. Edmund Egan—known by many as Ted—announced his retirement after more than 37 years with the company. Dr. Egan was one of five scientists that founded ONY Biotech, then ONY, Inc., in 1985 in hopes of developing a lung surfactant to treat premature infants born with respiratory distress syndrome, known as RDS. A ...
By ONY Biotech
-
CONVERT Clinical Trial Data Presented at ERS Demonstrates Early Success of AeriSeal System in Patients with Advanced COPD/Emphysema
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, announces the presentation of interim results from the CONVERT Study at the 2022 European Respiratory Society (ERS) International Conference. Data on the first 40 patients in the study demonstrated that treatment with the AeriSeal System successfully converted the ...
-
Nob Hill Therapeutics Awarded HHS Phase II Grant and Announces New CEO
ALBUQUERQUE, N.M. (PRWEB) August 23, 2022 Nob Hill Therapeutics (NHT), a VIC Technology Venture Development portfolio company, announced today that it has received an HHS phase II grant (up to $1.73M) to develop an inhaled therapy to treat lung fungal infection using an innovative first-of-its-kind dry powder nebulizer delivery platform technology. The funds will be used to develop the drug ...
-
Role of oxygen generator in the epidemic period of coronavirus disease
Why is the oxygen generator important? Oxygen is a life-saving therapeutic gas, which is often used to treat patients with severe pneumonia and other respiratory diseases. Oxygen generator is an electric medical equipment. It first inhales air, removes nitrogen, then generates a continuous oxygen source, and delivers concentrated oxygen to patients who need respiratory support in a controlled ...
-
Polarean Announces NDA Submission of 129 Xenon Gas MRI
Polarean Imaging plc (AIM: POLX), the medical-imaging technology company, with an investigational drug-device combination product for magnetic resonance imaging (MRI), announces its submission of a New Drug Application (“NDA”) and request for priority review to the US Food and Drug Administration (“FDA”) for hyperpolarized 129Xenon gas used to evaluate pulmonary ...
-
Medimprove – A New Private Clinic For Lung Revalidation With FRI
To address the need for better chronic respiratory care, Prof De Backer, emeritus Professor Pulmonology at the University of Antwerp and Chairman of The board of FLUIDDA NV, has opened a private clinic in Belgium near the Fluidda headquarters in Kontich, Belgium. The clinic uses a multidisciplinary approach for chronic respiratory failure. Care at this private clinic can involve exercise ...
By Fluidda
-
The Neotec Program finances a new transport ventilator with ICU benefits
INBENTUS , with name Iconic Solutions by Murcia SL, is carrying out a project co-financed with European funds from the Recovery and Resilience Mechanism and "Subsidized by the CDTI" supported by the Ministry of Science and Innovation within the NEOTEC PROGRAM , called " TRANSPORT RESPIRATOR WITH ICU BENEFITS ” SNEO-20211086 The project runs from January 1, 2022 to December 31, ...
By Inbentus
-
Nob Hill Therapeutics: Transforming Drug Delivery for Pulmonary Conditions
May 1, 2022 – Inhalation therapies, despite their prevalence in treating pulmonary diseases and respiratory-related conditions, have numerous setbacks, such as the deposition of aerosolized particles in the upper ...
-
CytoSorbents to Present at the Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum and Jefferies London Healthcare Conference
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces its participation in two upcoming investor conferences. CytoSorbents will present at the Canaccord Genuity Virtual MedTech, Diagnostics and Digital Health & Services Forum on Thursday, November 18, 2021. Dr. ...
-
CytoSorbents To Expand Direct Sales of CytoSorb to the United Kingdom, the Sixth Largest Medical Device Market in the World
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces that it has registered a new subsidiary, CytoSorbents Medical UK Limited, with plans to establish a direct sales presence within the United Kingdom (UK) and Ireland. CytoSorbents previously sold CytoSorb® via ...
-
Longeveron Enters into Cooperative Research and Development Agreement with the US Department of Veterans Affairs for its Phase 2 Alzheimer’s Trial
Longeveron Inc. (NASDAQ: LGVN) ("Longeveron" or "Company"), a clinical stage biotechnology company developing cellular therapies for chronic, aging-related and life-threatening conditions, announced today a Cooperative Research and Development Agreement (CRADA) with the United States Department of Veterans Affairs (VA) for the addition of a Miami VA clinical site to its ongoing Phase 2 ...
-
BioCardia Announces FDA Approval of Its IND for NK1R+ Mesenchymal Stem Cells for the Treatment of Patients Recovering from Acute Respiratory Distress due to COVID-19
BioCardia®, Inc. [Nasdaq: BCDA], a developer of cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today announced that the U.S. Food and Drug Administration (FDA) has approved the Company's Investigational New Drug (IND) application for BCDA-04, a proprietary allogeneic mesenchymal cell (MSC) population that is Neurokinin-1 receptor positive ...
-
CytoSorbents Receives Approximately $740,000 from the New Jersey Business Tax Certificate Transfer Program
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, today announced that it has received approximately $740,000, net of transactions costs, in cash proceeds from the sale of its 2020 Net Operating Loss (NOL) and R&D tax credits from the Technology Business Tax Certificate ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you