Biosensors International Group, Ltd. products
BioFreedom
BioFreedom - Model BA9 - Bridging Science and Innovative Technology
In the early 2000's the available mTOR inhibitor limus drugs, available for coronary stenting, were developed for systemic application. Biosensors developed its own proprietary limus technology by focusing during the early stages of drug development on its use for vascular technologies.
BioFreedom - Selectively Micro-Structured Surface (SMS)
Only the abluminal surface of the stent receives SMS treatment, allowing BA9™ to be contained within the micro-structured surface and delivered with high specificity to the vessel wall of the coronary lesions. The SMS process allows for an increased surface area for a uniform dose of BA9 to be delivered to the target lesion. With no need for polymer or carrier, BA9 and SMS make BioFreedom a true Drug-Coated Stent (DCS).
Powerline
Powerline - Model RISE™ NC - Balloon Dilatation Catheters
RISE™ NC’s deliverability and durability will accompany you in even the most challenging cases.
Biosensors - Model RISE SC - Angioplasty Balloon
The RISE SC angioplasty balloon from Biosensors International represents a sophisticated tool in lesion preparation, marked by significant advancements that cater to the needs of healthcare professionals. It is designed with an improved performance matrix, making it one of the most versatile options available on the market, available in over 70 size variations ranging from 1.00 to 5.0 mm. This product offers enhanced ease of use with features such as a reduced entry profile and new tip bonding processes, which together bolster its trackability and deliver a superior crossing profile. The new hydrophilic coating on the shaft improves maneuverability, while the higher nominal pressure aids in effective lesion preparation. Intended for use by licensed medical professionals, this device is part of Biosensors International's commitment to advancing interventional cardiology practices. However, it remains unavailable in certain regions including the United States.
BioMatrix Alpha
BioMatrix Alpha - Model Biolimus A9 - Proven Safety of the Biolimus Drug
BioMatrix™ Alpha presents the best in class stent platform design with unique pro-healing coating from the pioneer in abluminal biodegradable technology. It combines the proven safety of a DES with an abluminal biodegradable polymer, the proven efficacy of BA9™ and an innovative cobalt-chromium stent platform design.
Other Products
ALLEGRA - Model THV - Transcatheter Heart Valve for Aortic Stenosis
The ALLEGRA Transcatheter Heart Valve (THV) is engineered for treating severe calcified aortic valve stenosis, focusing on maintaining optimal hemodynamic performance with single-digit mean pressure gradients and expansive effective orifice areas. Designed for high-risk patients, the ALLEGRA leverages a unique self-expanding stent frame that promotes effective anchoring and sealing while allowing a flexible outflow for a wider valve opening. This design addresses the demands of both small annuli and valve-in-valve procedures ensuring minimal stress on leaflets, which translates to enhanced durability. Furthermore, TissGUARD technology aids in tissue protection by combining anti-calcification treatment with stress-reducing features. With a supra-annular valve position, it achieves physiological flow patterns that reduce gradients, platelet activation, and paravalvular leak, extending the valve's functionality. Additionally, its design maintains access to coronary ostia, preparing for potential future interventions.
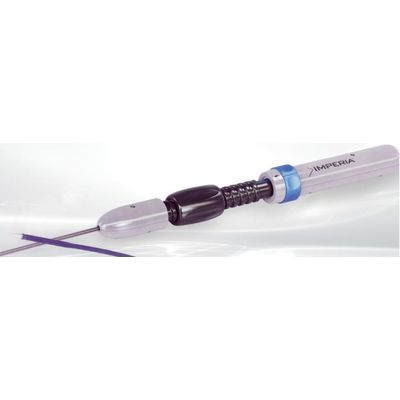
NVT IMPERIA - Delivery System for ALLEGRA Transcatheter Heart Valve
The IMPERIA Delivery System, a solution by NVT GmbH, is specifically engineered for the efficient transfemoral implantation of the ALLEGRA Transcatheter Heart Valve. This advanced system is distinguished by its capacity for precise commissural alignment, a critical factor in successful valve implementation. Enabled by six radiopaque markers, the system facilitates the accurate alignment of the valve during the deployment process. Among its notable features is the ability to achieve up to 80% valve deployment with the option for recapture, repositioning, or full retrieval if needed, thus ensuring predictable outcomes without valve movement upon detachment. The system is linked with various studies that have evaluated its clinical performance and hydrodynamic function. The ALLEGRA device and IMPERIA System bear CE approval, affirming their compliance with European health and safety standards. Commercialization of these medical devices is supported by collaboration between NVT GmbH and Biosensors International Group, with distribution limited to regions where healthcare authorities have granted product registration.
NVT ALLEGRA - Model TF - Transcatheter Heart Valve with IMPERIA Delivery System
The ALLEGRA Transcatheter Heart Valve integrated with the IMPERIA Delivery System offers a state-of-the-art solution for transcatheter aortic valve interventions. Designed to maintain hemodynamic stability throughout deployment, the Permaflow Delivery System allows for continuous blood flow, providing medical professionals with the time needed to accurately position the device without stress. The system features a straightforward three-step deployment process: initial flow maintenance, inflow release, and final detachment of the valve. ALLEGRA is recognized for its occlusion-free deployment technique, minimizing risks associated with hemodynamic compromise. The device's novel self-expanding functionality, alongside its CE approval, underscores its compliance with rigorous safety and efficiency standards. The IMPERIA system is fully repositionable, enhancing the precision of implants in complex cases such as severe calcific aortic stenosis or degenerated surgical bioprostheses, contributing to improved patient outcomes.
Biosensors - Model BioMC - Clinically Proven Microcatheter for Interventional Cardiology
The BioMC™ microcatheter, developed by Biosensors International Group, is designed specifically for interventional cardiology procedures. Its architecture features a soft, tapered and radiopaque tip, eliminating the need for additional markers. The catheter is optimized to strike a balance between visibility, durability, and inner diameter. It incorporates a PTFE liner with braiding for enhanced pushability, coupled with coiling to improve flexibility. The tailored stiffness gradient from proximal to distal ends aids in effective force transmission and support. The large inner shaft diameter contributes to a streamlined crossing profile, facilitating smooth guidewire exchange. Despite its advanced design, the device's use is restricted to sale and operation by physicians, highlighting the need for professional healthcare oversight. This product is not available for sale in the United States and a number of other regions, pending appropriate health authority registrations and approvals.