Zhejiang Runlab Technology Co., Ltd products
Liquid Handling - Serological Pipettes
Runlab - Polystyrene Gamma Irradiation Serological Pipettes
Manufactured from virgin polystyrene are ideal for accurate liquid transfer or mix. Management system EN ISO13485 with SOPs and 100,000 grade clean-room guarantee quality. Available with 6 capacity of 1.0, 2.0, 5.0, 10.0 , 25.0 and 50.0ml The 1ml, 2ml and 5ml are a one piece construction and the 10ml, 25ml and 50ml pipets are made with a durable ultrasonically welded 3 piece construction. Clear black printing guarantee maximum an accuracy of ±2%.
Specimen Collection - Specimen Containers
Runlab - Model 5.3OZ/160ml - Histology Specimen Container
Made of Non-Cytotoxic Polypropylene. Cap Material: HDPE. Ideal for liquid specimen collection, transportation and storage in harsh conditions. To Eliminate formalin leakage and evaporation. Top and bottom gripping assures easy opening and closing when wearing gloves. Half-transparency containers with graduations have easy viewing. Graduated on three sides. Meets 95Kpa Transportation Guideline. MAX capacity till edge of mouth: 188mL.
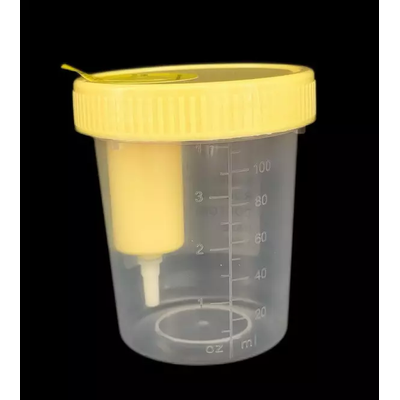
Runlab - Model 120ml - Vacuum Urine Container
Mainly used for pollution-free operation before urine specimen collection and inspection. Cup Material : PP. Good sealing performance ensures the pre-analytical quality control and provides a guarantee for the standardization of sample testing.
Specimen Collection - Transport Tubes
Runlab - Model Ø16xH100mm, 10ml - Transport Tubes
Transport Tubes, Screw Cap, Free-standing Bottom, Ø16xH100mm, 10ml. Manufacturered from virgin polypropylene. 1-1/4 turn thread design for super fast screwing, Printed Graduation. Meets 95Kpa transportation guide-line.
Specimen Collection - Transport Swabs
Runlab - Transport Swabs Without Medium
Manufactured from virgin polypropylene or LDPE tubes,round bottom with size φ13mmxH150mm. Ideal for sample collection and transport device for bacteria. CE marked as Clas lla in accordance with the medical Device Directive 98/79/EC. Color coded caps for easier identification.