Medical Testing News
-
Protheragen-ING Lab, a Trustworthy GLP Service Provider, Unveils Its Cutting-Edge Medical Device Services
Protheragen-ING Lab, a well-known and reputable Good Laboratory Practices (GLP) service provider, has recently unveiled its cutting-edge medical device services. With a track record of excellence and reliability in providing high-quality services to the pharmaceutical and biotechnology industries, Protheragen-ING Lab is now expanding its offerings to include comprehensive solutions for medical ...
-
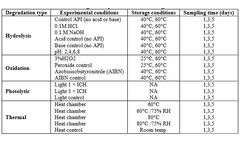
CD Formulation Enables Efficacy Evaluation of Oral Thin Films: Ensuring Therapeutic Efficacy and Safety
A breakthrough in CD formulation has enabled researchers to evaluate the efficacy of oral thin films, ensuring both therapeutic efficacy and safety. This new development has the potential to revolutionize the way oral medications are tested and administered, leading to more precise and effective treatments for a wide range of medical conditions. The development of oral thin films has gained ...
-
STEMart Launches New Force Degradation Services to Ensure Drug Stability and Safety
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and efficacy throughout their shelf life. Forced ...
By STEMart
-
STEMart Launches Bioburden and Sterility Testing for Medical Devices
STEMart, a U.S.-based provider of comprehensive services for all stages of medical device development, has recently introduced the Bioburden and Sterility Testing services for medical devices under the guidance of the ISO 11731 method. For medical device manufacturers, sterility assurance is a vital part in producing safe products. The FDA and other regulatory agencies require validation of ...
By STEMart
-
ViGeneron Announces Closing of Series A Financing to Drive Development of Next Generation Gene Therapy Pipeline
ViGeneron GmbH, a gene therapy company, announced the closing of its series A financing round led by WuXi AppTec and Sequoia Capital China. The proceeds will enable ViGeneron to accelerate its proprietary viral vector-based gene therapy platforms and drive product development in its two lead ophthalmic gene therapy programs. Funds will advance development of ViGeneron’s next-generation ...
-
Modular Medical Announces Product Testing Update
Modular Medical, Inc. (the "Company" or "Modular Medical") (NASDAQ:MODD), a development stage, insulin delivery technology company seeking to launch the next generation of easy to use and affordable insulin pump technology, today announced an update on the development of its initial insulin pump product. The Company has continued to make progress on the testing required for its 510(k) submission ...
-
Enterprise Forum
The 101 Corridor is leading the way in new diagnostics that will bring information to you and your doctor within minutes or hours. You will no longer have to wait for days for a central diagnostic laboratory to provide critical medical test data regarding your health. This program showcases a range of young diagnostic companies that are poised to launch exciting products and services that will ...
-
Cingulate Announces Agreement with Societal CDMO and Provides Clinical Update
Cingulate Inc. (NASDAQ: CING), a biopharmaceutical company utilizing its proprietary Precision Timed Release™ (PTR™) drug delivery platform technology to build and advance a pipeline of next-generation pharmaceutical products, today announced it has executed a Master Services Agreement (MSA) with Societal CDMO, Inc. (NASD: SCTL), a bi-coastal contract development and manufacturing ...
By Cingulate
-
STEMart Launches Biological Indicator Sterility Testing Service for Medical Devices
STEMart, a U.S.-based provider of comprehensive services for all stages of medical device development, has recently introduced Biological Indicator Sterility Testing services for the industry of medical devices. This testing is performed according to the exposure of Biological Indicators (BIs) after completion of the sterilization load, and it is a qualitative test to reveal results that can ...
By STEMart
-
XPhyto Signs Letter of Intent for US Manufacturing and Strategic Business Opportunities
XPhyto Therapeutics Corp. (CSE:XPHY / OTC:XPHYF / FSE:4XT) (“XPhyto” or the “Company”) is pleased to report that it has signed a non-binding Letter of Intent (“LOI”) to identify and assess potential business synergies for manufacturing, import/export, distribution and product development with a US-based thin film manufacturing firm (the “Firm”). ...
-
XPhyto Pursues Potential Application of its Oral Dissolvable Biosensor Inflammation Test for Buprenorphine-related Dental Disease
XPhyto Therapeutics Corp. (CSE:XPHY / OTC:XPHYF / FSE:4XT) (“XPhyto” or the “Company”) is pleased to report a significant potential market opportunity for its oral dissolvable (“ODF”) biosensor screening tests for oral inflammation. Certain buprenorphine medicines prescribed to treat opioid use disorder (“OUD”) and pain have been recently associated ...
-
How Do We Fill The Data Gap In Virtual Care?
Telehealth use skyrocketed in the early months of the Covid-19 pandemic when many health providers were forced to deliver services remotely. Although telehealth visits have dropped from their peak in 2020, they remain elevated even as many patients return to in-person care, according to the results of an analysis by Epic Research and the Kaiser Family Foundation. Survey results show that ...
-
Why Innovation in Blood Testing Matters
Why should payers care about clinical laboratory testing? Clinical laboratory testing plays an essential role in the delivery of health care. From early detection and diagnosis of disease to individualized treatment plans based on a person’s unique biology, medical care depends on accurate and timely health data. Seventy percent of medical decisions depend on lab test results, so making ...
-
CONVERT Clinical Trial Data Presented at ERS Demonstrates Early Success of AeriSeal System in Patients with Advanced COPD/Emphysema
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, announces the presentation of interim results from the CONVERT Study at the 2022 European Respiratory Society (ERS) International Conference. Data on the first 40 patients in the study demonstrated that treatment with the AeriSeal System successfully converted the ...
-
Babson Diagnostics and Pharmacy Podcast Network Announce Partnership That Will Take Listeners Inside Largest Retail Pharmacy Trade Show
Babson Diagnostics, a science-first, health care technology company, today announced a media partnership with the Pharmacy Podcast Network in connection with the 2022 National Association of Chain Drug Stores’ Total Store Expo, the largest gathering of retailers and suppliers in the health and wellness industry. As part of the partnership, the Pharmacy Podcast Network, the most popular ...
-
STEMart Introduces New Antibiotic Potency Tests for Medical Devices
STEMart, a provider of CRO services dedicated to integrated medical device and diagnostic clinical development, introduced new Antibiotic Potency Tests to assess the bioactivity or potency of various antibiotics for medical devices. The development of these new tests strictly follows the Antibiotics-Microbial Assays in USP General Chapter 81 to ensure the highest efficacy. Microbial potency ...
By STEMart
-
Diasorin launches Liaison MeMed BV, the first high throughput blood test to differentiate between bacterial and viral infections, in countries accepting the CE Mark
Liaison Memed Bv is an immune system based protein signature test for distinguishing between bacterial and viral infections The test was developed following the licensing agreement signed with Memed as announced in September 2020 The new test is available on the Liaison Xl platforms in countries that accept CE mark and U.S. food and drug administration clearance is planned for 2022 ...
By MeMed
-
STEMart Launches Microbiology & Sterility Testing Services for Medical Devices
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, introduces comprehensive microbiology and sterility testing for sterile, non-pyrogenic products. STEMart has extensive expertise in microbiology as well as sterility testing and remains dedicated to a full-service experience that supports manufacturers in meeting ...
By STEMart
-
Chiricahua Community Health Centers, Inc., Launches STI Testing Program for Women with Same-Day Results
The Body Agency Collective, Visby Medical, Inc. and CCHCI observe National STD Awareness Month with women’s health initiative on US-Mexico border. CCHCI among first healthcare providers in the nation to adopt new handheld PCR technology to detect gonorrhea, chlamydia and trichomoniasis in a single visit. Chiricahua Community Health Centers, Inc. (CCHCI) today announced a collaboration ...
-
STEMart Launches Pyrogenicity Testing Service for Medical Devices
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, now launches Pyrogenicity Testing service for the medical device industry. This new testing follows the biocompatibility guidelines modified for medical devices. Due to the risks associated with medical devices, comprehensive medical device testing throughout the product ...
By STEMart
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you