Good Manufacturing Practices (Biopharmaceuticals) Equipment
-
PremiumManufactured by Powder Systems Limited (PSL)based in UNITED KINGDOM
The LabMSR system is a GLP piece of equipment that has been developed to enable global drug manufacturers to generally replicate, at R&D scale, some of the process benefits of PSL’s full scale production MicroSphere Refiners (MSR). Due to their characteristics and properties, polymeric microspheres have traditionally been ...
-
PremiumManufactured by Powder Systems Limited (PSL)based in UNITED KINGDOM
The MSR™ MicroSphere Refiner is a cGMP piece of equipment that allows drug developers and manufacturers to complete a wide range of aseptic processes with their microspheres at various scales. It is a one-of-its-kind solution has been developed and refined over decades following a Quality-by-Design (QbD) approach, taking into ...
-
Manufactured by Kopran Ltd. & Kopran Research Laboratories Ltd.based in INDIA
Conformation to the Good Manufacturing Practices of International Standards. Approved by 11 countries including TGA Australia,MCC South Africa, MHRA UK,FMHACA Ethiopia, PPB Kenya,MOH Oman,MOH Yemen, NDA Uganda, MOH Ivory Coast GMP Certification by FDA, Government of Maharashtra. WHO GMP Certification by CDSCO, Government of India More than ...
-
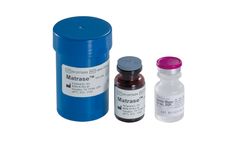
Manufactured by InGeneron, Inc.based in USA
InGeneron’s proprietary MatraseTM Reagent is a high purity enzymatic mixture for the release of mesenchymal stem cells and other regenerative cells from adipose tissue. The product is manufactured under a contract by a major biopharmaceutical company under current Good Manufacturing Practices (cGMP) and tested by ...
-
Manufactured by 3DBio Therapeuticsbased in USA
GMPrint™ technology is a purpose-built therapeutic-grade 3D-bioprinter, designed and manufactured by 3DBio. Research-grade systems do not meet key requirements set by the FDA for manufacture of therapeutics, broadly referred to as current Good Manufacturing Practice (cGMP). GMPrint™ comprises ...
-
Manufactured by SOL Spabased in ITALY
production processes in the whole of Europe the Good Manufacturing Practice rules, in conformity with Directive 91/356/EEC. The SOL Group boasts an over 50 year-long experience in the supply of MEDICINAL GASES to the public and private hospital field. Over the years, this partnership has identified the new needs of the industry to which SOL ...
-
Manufactured by BioMaxima SAbased in POLAND
surfaces is recommended in the ISO standards 14698-1 (2), ISO 18593 (3), USP chapter 1116 (4) and in Good Manufacturing Practice ...
-
Manufactured by KASAG Swiss AGbased in SWITZERLAND
out non-destructive testing on the finished products before acceptance. In addition, we prepare all necessary documents with respect to GMP (Good Manufacturing Practice) requirements and for the validation and qualification procedures (DQ, IQ, OQ, PQ) for the manufacture of pharmaceutical and biotechnology ...
-
Manufactured by Spire Integrated Solutionsbased in USA
These sterilizers are designed for all Healthcare applications including surgery, central sterile supply and clinical laboratories. PRIMUS sterilizers are designed and manufactured in the USA according to Quality Management Systems which are in compliance with ISO 9001:2008, ISO 13485:2003, CMDCAS and FDA's Good Manufacturing ...
-
Manufactured by Ciseobased in BELGIUM
Sterile filtration of vaccines and parenteral drugs before the filling step is an important requirement in terms of good manufacturing practices in the pharmaceutical sector. You want to make sure that it is carried out under the safest conditions: filter-integrity tests before and after production, sterility assurance and production ...
-
Manufactured by Keller Technology Corporation (KTC)based in USA
. We follow Current Good Manufacturing Practices (cGMP), and our facilities are ISO 9001 and ISO 13485 certified, so we have the qualifications and experience to provide contract manufacturing services for your medical capital equipment product. From design and engineering to production and supply chain management, KTC is not ...
-
Manufactured by Toppy Srlbased in ITALY
Toppy Pharma Advance WB pallet changer can lift and turn loads up to 1000 Kg. The max. possible height of the load is 1550 mm (pallet included). Like any Toppy equipment, Toppy Pharma Advance WB complies with the strictest European and American rules and pays special attention to the GMPs, with its smooth surface and bevelled shape assuring the utmost hygiene and cleanliness. Toppy Pharma Advance ...
-
Manufactured by General Machine Products (KT), LLCbased in USA
The latest word in aerial cable lashing. Perfect for all telcom cable construction, including Coaxial and FTTx cable. With its light weight and parallel-pull capability, you’ll string cable quickly and easily, over all outside plant cable routes. From the company that pioneered the modern automatic lasher with Western Electric back in the 1940’s…. comes Apollo… a machine ...
-
Manufactured by Toppy Srlbased in ITALY
Toppy Pharma Advance pallet changer can lift and turn loads up to 1000 Kg. The max. possible height of the load is 1550 mm (pallet included). Like any Toppy equipment, it complies with the strictest European and American rules and pays special attention to the GMPs, with its smooth surface and bevelled shape assuring the utmost hygiene and ...
-
Manufactured by Cleangrad d.o.o.based in SLOVENIA
Aluminum (ALU) or laminate (HPL) face wall claddings consist of modular panels, which are mounted on the base wall structure with special profile elements. Face wall claddings appear similar to Cleangrad’s wall panels and provide the same technical characteristics. Joints between the panels are sealed with special silicone, meeting all requirements of internationally accepted GMP and FDA ...
-
by Pureims B.V.based in NETHERLANDS
In addition to in-house programs, we aim to co-develop products with partners who are pursuing NCEs and/or repurposed medicines for local or systemic application. In this respect, PureIMS collaborates with several companies. PureIMS has GMP facilities for the clinical-scale manufacturing of Cyclops™-based products. The ...
-
Manufactured by Cleangrad d.o.o.based in SLOVENIA
Cleangrad’s Kerrock partition walls C-W-KERROCK consist of modular panel elements, which by its kerock final layer combines beauty of ceramics and durability of natural stone. Kerrock partition wall systems are fully flexible and allow a separate de-installation of each individual panel in a very short time. Joints between the panels are sealed with special silicone, meeting all ...
-
Manufactured by Sampling Systems Ltd.based in UNITED KINGDOM
Our range is both varied and extensive. To ensure you get the correct sampler for your application, please contact Sampling Systems here and we will be happy to discuss your requirements and suggest a solution or send you a full range ...
-
Manufactured by Aldevron, LLCbased in USA
In Stock, Royalty-Free, Helper Plasmid Solution for Recombinant AAV Manufacturing. Triple transfection is the most common method for rAAV production and does not require co-infection with adenovirus; a helper plasmid provides adenoviral genes. Aldevron's pALD-HELP has several advantages: Immediately available at research grade and ...
-
Manufactured by Aldevron, LLCbased in USA
In Stock, Royalty-Free, Helper Plasmid Solution for Recombinant AAV Manufacturing. Triple transfection is the most common method for rAAV production and does not require co-infection with adenovirus; a helper plasmid provides adenoviral genes. Aldevron's pALD-HELP has several advantages: Immediately available at research grade and ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you