Medical Drugs Software
-
Manufactured by CENTOGENE N.V.based in GERMANY
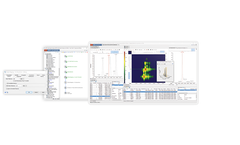
Analyze, interpret, and report genomic variants with our decentralized diagnostic solution – no matter where you are located. CentoCloud is a cloud-based Software as a Service (SaaS) platform specifically designed for the rapid bioinformatic analysis and interpretation of Next-Generation-Sequencing (NGS) data based on CENTOGENE’s proprietary Biodatabank and external reference ...
-
by Genedata AGbased in SWITZERLAND
Genedata Expressionist® is an open and scalable enterprise software whose unique workflow-based approach enables custom-built solutions that mirror your existing processes, address your specific business and technical needs, and transform your raw data into actionable insights. ...
-
based in USA
QDOSE® Multi-purpose Voxel Dosimetry (Personalized Dosimetry in Molecular Radiotherapy) provides state-of-the-art assessment of radiation absorbed dose to organs and tissues of the body from medically administered radiopharmaceuticals. This software is a complete, one-stop solution for all internal dosimetry needs with multiple parallel ...
-
based in CAMBODIA
Qdata™ by Verana Health are disease-specific, fit-for-purpose data modules designed to confidently drive business insights and rigorously inform research outcomes. Qdata spans three therapeutic areas-ophthalmology, neurology, and urology—and reflects deep patient journeys across robust demographics. Qdata helps to unlock quality research insights along the entire drug and medical ...
-
based in USA
Maintaining accurate and reliable electronic health records is one of the primary goals for Oasis Technologies Group. It can be a daunting and burdensome task maintaining and managing client records. When insurance companies audit records you may become stressed out and overwhelmed. Many times the paperwork and record maintenance responsibilities just gets in the way of providing effective and ...
-
Manufactured by Intelligencebased in GERMANY
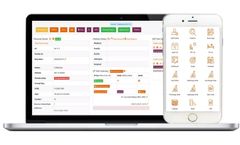
Ontosight Influence is an AI-based, one-stop solution for identifying and managing top and emerging key opinion leaders (KOLs) across the entire drug discovery and development value chain, from the preclinical phase to postlaunch support. How do you find the right KOLs online in your therapeutic area of interest. How do you know which KOL can help in research and clinical development of your ...
-
by MocDocbased in INDIA
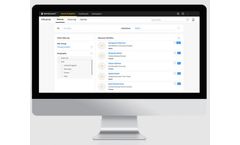
Centralize patient information for effortless access, updates, and analysis by medical professionals. MocDoc Clinic Software is designed to help even the busiest clinics run more efficiently with a fully integrated Clinic management system and Practice ...
-
Manufactured by Synbiosisbased in UNITED KINGDOM
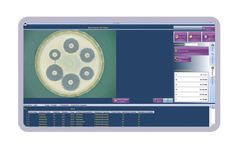
The revolutionary eAST software is designed for use with ChromaZona and ProtoCOL 3 automated zone measurement systems. The software quickly and simply measures zones around antibiotic discs and calculates antibiotic resistance from results to help guide treatment options. At the touch of a button the results can be automatically compared to data from all the tested organisms that have breakpoint ...
-
based in USA
The leading global regulated Digital Health platform for biopharma and ...
-
Manufactured by ARANZ Medical Limitedbased in NEW ZEALAND
Silhouette delivers the data accuracy you need to prove your endpoints. 3D depth measurements. Consistent, clear images. Accurate, reliable data across users. Infection ...
-
by PathAI, Inc.based in USA
AISightTM is a web-based digital pathology slide viewing platform that enables the management and analysis of whole slide ...
-
based in USA
The medical device reporting (MDR) requirements of 21 CFR 803 apply to manufacturers, importers, distributers, and device user facilities in the United States. Such facilities are required to report adverse device-related events to the Food and Drug Administration (FDA) or the device manufacturer, and occasionally to both. These requirements are supplemental to the quality system requirements of ...
-
based in USA
Are you tired of submitting your billing claims and getting denials? Is it time consuming and distracting you from day to day operations of your agency. At Oasis Technologies, we have teamed up with some of the most respected revenue cycle management companies in the industry to provide you with choices and billing options. We want to help you reduce the cost and stress of billing so you can ...
-
Manufactured by Getinge ABbased in SWEDEN
T-DOC Select is Getinge's traceability and sterile supply management solution suited for mid-sized hospitals. T-DOC Select helps hospitals optimize the sterile production and enables regulatory compliance for sterile reprocessing. T-DOC Select provides uncompromising quality assurance by guiding, documenting, and tracking all processes in the sterile production. T-DOC Select provides a fully ...
-
based in USA
Cell Line Select® is Champions' online, searchable database containing over 100 Human cancer cell lines, including characterization datasets such as gene mutations identified through Whole Exome Sequencing (WES) and gene expression levels quantified using RNASeq technology. ...
-
Manufactured by Caresyntaxbased in GERMANY
Rescale is a cloud-based platform for computational engineering and R&D. Makes sharing simulation insights seamless, flexible and automated. Integrates with SPDM to capture the intention of every ...
-
by Instem Groupbased in UNITED KINGDOM
Provantis is a modern, fully integrated Windows-based system for organizations and universities engaged in non-clinical evaluation studies. From single-user Pathologists to full-function global Toxicology/Pathology laboratories, Provantis streamlines processes and workflows with straightforward, intuitive functionality for simple and complex studies within a GLP or non-GLP ...
-
based in INDIA
A comprehensive and accurate medicine database for different healthcare sectors. Certified database approved by healthcare experts and practitioners. What is a medicine database? To make it short and simple, a medical database has medical information that refers to information such as drugs used to treat a patient caused by some illness or disease. Basically, it is full-fledged data related to ...
-
Manufactured by Codonicsbased in USA
Codonics award-winning Safe Label System (SLS) is the world’s leading solution for safe, compliant, and fast medication labeling — anywhere medications are prepared! An FDA Class 2 medication safety system, SLS uses barcode technology to improve safety, compliance and efficiency during medication preparation. SLS helps to eliminate vial and ampoule swaps, mislabeling, and syringe ...
-
Manufactured by OpGen, Inc.based in USA
Acuitas Lighthouse is the first cloud-based software to identify, track, and predict antibiotic-resistant infections based on genetic information. It is being developed to help guide care and suggest the most effective medications for infected ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you