Blood Donor (Blood Collection) Equipment
-
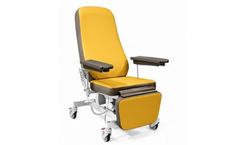
Manufactured by Malvestio Spabased in ITALY
Fully electric Blood-donor chair with variable height. Multifunction push-button handset with height, backrest, legrest and Trendelenburg adjustment. Rotating and height adjustable shaped armrests. 4 wheels Ø 100 mm with brake. Safe working load 200 kg. ...
-
Manufactured by Trinity Biotech Plc.based in IRELAND
The Captia™ Malaria Total Antibody EIA intended for use by appropriately trained and qualified personnel for the detection of antibodies to P.falciparum, P.vivax, P.ovale and P.malariae in human serum and ...
-
Manufactured by Trinity Biotech Plc.based in IRELAND
The Captia Malaria Total Antibody EIA is intended for use by appropriately trained and qualified personnel for the detection of antibodies to P.falciparum, P.vivax, P.ovale and P.malariae in human serum and ...
-
Manufactured by Trinity Biotech Plc.based in IRELAND
The Captia™ Syphilis Total Antibody EIA kits is intended for use by appropriately trained and qualified personnel for the detection of antibodies to Treponema pallidum in human serum and plasma. The product may be used for the screening of blood donors, the screening of pregnant women, and to aid in the diagnosis of patients where ...
-
Manufactured by Tempo Medical Productsbased in USA
Lightly textured to provide maximum control; Innovative design prevents tourniquet from slipping or unraveling; Recommended for single patient use, to help reduce the risk of cross-contamination; Used by phlebotomists to initiate blood draws and begin the apheresis process; Ideal product for blood donor and IV starter ...
-
Manufactured by Polymedicurebased in INDIA
Coated with micronised silica which acts as clot activator. Suitable for clinical serum and blood donor screening chemistry ...
-
Manufactured by Tempo Medical Productsbased in USA
The non latex material will safeguard patients by eliminating latex related allergic reactions; Innovative design prevents tourniquet from slipping or unraveling; Used by phlebotomists to initiate blood draws and begin the apheresis process; Ideal product for blood donor and IV starter kits; Conveniently packed flat for ease of use and ...
-
Manufactured by Polymedicurebased in INDIA
Coated with micronised silica which acts as clot activator and a gel is added that seperates blood cells from serum. Suitable for clinical serum and blood donor screening chemistry ...
-
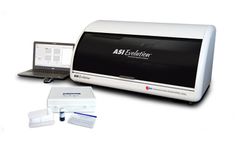
Manufactured by Arlington Scientific, Inc.based in USA
FDA Cleared For Diagnostic, Blood Donor and Cadaveric Tissue Screening; For over 37 years Arlington Scientific has been a pioneer in delivering new products and technologies to meet the growing demands of clinical diagnostics, blood donor and cadaveric tissue laboratories worldwide. The ASI Evolution® Automated Syphilis ...
-
Manufactured by EKF Diagnostics USAbased in USA
The UltraCrit™ Hematocrit Device is a CLIA waived hematocrit device, high accuracy hematocrit measurement for blood donor screening. EKF Diagnostics UltraCrit™Hematocri device is a point-of-care hematocrit device delivering highly accurate measurements for blood donor screening. UltraCrit™Hematocrit Device is a ...
-
Manufactured by Ortho Clinical Diagnosticsbased in SINGAPORE
We share the vital mission of donor screening centers and labs – to enable reliable and timely access to safe blood products for patients every day. Deliver safe, reliable blood while facing today's challenges. Blood donor centers balance a highly regulated environment with the persistent expectation to do ...
-
Manufactured by Arlington Scientific, Inc.based in USA
Arlington Scientific’s Automated RPR Syphilis Test is FDA cleared for diagnostic, blood donor screening and cadaveric RPR testing, for use on the ASI Evolution® Automated Syphilis Analyzer. Nontreponemal tests, such as the ASI Automated RPR test, are more reliable indicators of untreated syphilis infections and are used to ...
-
Manufactured by Access Biobased in USA
The CareStart™ EZ HIV 1/2 is an in vitro RDT for the qualitative detection of antibodies to HIV type-1 and/or type-2 in serum, plasma, capillary whole blood or venous whole blood. The device is a single-use intended for healthcare professional use as an aid to diagnosis only – not for screening blood ...
-
Manufactured by InTec PRODUCTS, INC.based in CHINA
of blood donors and for aid in the diagnosis of clinical conditions related to infection with HIV-1 and/or HIV-2. ...
-
Manufactured by Cellphire Therapeutics, Inc.based in USA
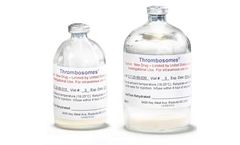
Thrombosomes are made by pooling platelet units from 10 Group O (Universal) blood donors, removing the plasma and adding a natural sugar, trehalose, to stabilize the cells. Water is then removed through a freeze-drying process resulting in a dry powder. The dry product is heat treated as a final step to reduce the risk of viral ...
-
Manufactured by BMS K Groupbased in SPAIN
The blood bag tube stripper is a “plier-like” tool, intended to be used with PVC blood tubing, to strip undiluted blood from the donor tubing. The blood bag tube stripper is easy to use and highly ergonomic. It can be used with different sizes of blood bag tubes due to the screw ...
-
Manufactured by Avioq, Inc.based in USA
The Avioq HIV-1 Microelisa System is intended for use as an aid in diagnosis of infection with HIV-1. It is not intended for use in screening blood donors. Developed as a standard two-step indirect microelisa system assay, the Avioq HIV-1 Microelisa System contains coated 96-well microwell plates, color-coded, liquid calibrator and ...
-
Manufactured by Sisu Global Healthbased in USA
The Hemafuse is a handheld, mechanical device for intraoperative autotransfusion of blood collected from an internal hemorrhage, meant to replace or augment donor blood in emergency ...
-
Manufactured by SYMATESEbased in FRANCE
of fibrin. In vitro tests were conducted on the blood of 3 donors to evaluate the performance of HEMOTESE® compress compared to a reference product made of oxidized regenerated cellulose. ...
-
Manufactured by Advanced Cellular Dynamics, Inc. (ADC)based in USA
Our catalog of primary immune effector cells derive from whole blood obtained from anonymous donors. Individual subsets are affinity purified by negative selection when possible, or positive selection when necessary. The methods deployed are disclosed within the specification data so that you can decide if the process used might adversely ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you