Cardiac Devices Equipment For Medical Equipment
-
Manufactured by Allergan Aestheticsbased in USA
Uses: The CoolTone® device is FDA-cleared for improvement of abdominal tone, strengthening of the abdominal muscles, and development for firmer abdomen. CoolTone® is also FDA-cleared for strengthening, toning, and firming of buttocks and ...
-
Manufactured by RayBiotech Life, Inc.based in USA
The Cytokine Array C5 is a membrane-based antibody array for screening and comparing expression levels of 80 human cytokines in any liquid sample ...
-
Manufactured by Elixir Medical Corporationbased in USA
The DESyne BD Novolimus-Eluting Coronary Stent System represented a significant advancement in biodegradable coating technology, eliminating the need for a primer coating and achieving a bare metal surface within six to nine months, while providing excellent DES ...
-
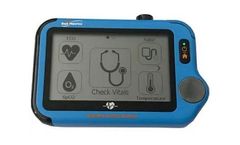
Manufactured by BodiMetricsbased in USA
BodiMetrics™ VitalsRx captures all of your vital signs in less than a minute while also providing continuous ECG (24 hour Holter) monitoring, recording and reporting. Also, monitor, record and report on Blood Oxygenation (SpO2 and Pulse Rate for up to 15 hours). Combined VitalsRx provide easy cost-effective monitoring and screening for heart irregularities and sleep related health issues ...
-
based in USA
The TORVAD is a Toroidal Ventricular Assist Device. VADs are fully implantable blood pumps that increases cardiac output for patients with heart failure. The TORVAD produces pulsatile ejections with two independently controlled pistons. One piston acts as a virtual valve while the other rotates around the toroidal pumping chamber and simultaneously aspirates and ejections blood by ...
-
Manufactured by Utah Medical Products, Incbased in USA
The Liberty® System is an easy-to-use pelvic floor stimulation system for effective non-surgical treatment of urinary incontinence. It is designed to treat stress, urge and mixed incontinence, and consists of a stimulation device and a choice of three comfortable ...
-
Manufactured by Artivion, Incbased in USA
Type A Aortic Dissection (TAAD) presents itself emergently. Left untreated, mortality of type A dissection is reported to be approximately 1% to 2% per hour after onset of symptoms1 and can lead to 50% mortality in the first 48 ...
-
Manufactured by Sartorius CellGenix GmbHbased in GERMANY
CellGenix® Recombinant Human EGF reliably promotes the expansion of mesenchymal stem cells (MSCs). It in addition promotes the differentiation of embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs)-derived hepatoblasts. CellGenix® rh EGF is produced in our dedicated animal-free facility ensuring maximum safety for optimal use in ATMP ...
-
Manufactured by Atacor Medical, Inc.based in USA
First of its kind, the innovative AtaCor EV Temporary Pacing System is advancing the standard of cardiac pacing. It is the only implantable temporary pacing system designed to leave the heart and vascular system completely untouched—for prompt pacing support without the discomfort, risk, and limitations of traditional ...
-
based in USA
Developing a low-profile, high-flow percutaneous ventricular assist device (pVAD) for high risk coronary intervention and cardiogenic ...
-
Manufactured by Berlin Heart GmbHbased in GERMANY
EXCOR® Pediatric is a mechanical, pulsatile heart support system (Ventricular Assist Device, VAD). The paracorporeal VAD is used for the short to long-term support of the left and/or right ventricular pumping function. It is indicated for children with life-threatening heart failure after all conservative therapeutic options have been exhausted. EXCOR® Pediatric is designed for use in ...
-
Manufactured by Stron Medical, Part of Q3 Medical Groupbased in GERMANY
The GALAXY Rapamycin-Eluting Coronary Stent System is the first carbonized stent (Inert Carbon Technology) with a completely biodegradable polymer coating which contains Rapamycin (Rapasorb™) as a highly effective drug for preventing thrombotic and re-stenotic ...
-
Manufactured by Tsunamed, Part of Q3 Medical Groupbased in GERMANY
The MAGMA Rapamycin-Eluting Coronary Stent System is the first carbonized stent (Inert Carbon Technology) with a completely biodegradable polymer coating which contains Rapamycin (Rapasorb™) as a highly effective drug for preventing thrombotic and re-stenotic ...
-
Manufactured by Artivion, Incbased in USA
Homografts, such as CryoValve Aortic Valves, are recommended by the current STS Clinical Practice Guidelines3 for patients with extensive active endocarditis destruction of the aortic annulus (Class I, Level of Evidence B) and can be considered for patients with endocarditis without annular destruction, especially when the potential for reinfection is elevated. (Class IIa, Level of evidence ...
-
Manufactured by Relisys Medical Devices Limitedbased in INDIA
Highlighting Features: • Premier+ is an improved version of our first indigenously developed Sirolimus Eluting Coronary Stent System • Premier+ is with unique open and closed cell stent design on L 605 Cobalt Chromium stent platform with strut locking mechanism. This offers excellent radial strength and minimum recoil • The strut thickness for Premier+ is 63 μm • The delivery system in Premier+ ...
-
Manufactured by EntroGen, Inc.based in USA
The KRAS gene encodes a small GTPase that plays a key role in transducing signals from the epidermal growth factor receptor (EGFR) to downstream effectors. KRAS mutations have been commonly found in several types of human malignancies, such as metastatic colorectal cancer (mCRC), lung adenocarcinoma and thyroid cancer. The most common mutations are found in codons 12, 13 and 61. Several studies ...
-
Manufactured by InSphero AGbased in SWITZERLAND
Test efficacy of anti-NASH drugs in an advanced 3D human liver disease model. The InSphero 3D InSight™ Human Liver NASH Model consists of an advanced microtissue co-culture of healthy primary human liver cells combined with specially formulated media and cocktails that induce the progression of NAFLD and NASH from steatosis to liver inflammation and fibrosis. Ideal for investigating ...
-
Manufactured by W. L. Gore & Associatesbased in USA
With more than 20 years of experience, the trusted performance of the GORE EXCLUDER Device is paired with the intuitive GORE C3 Delivery System to provide optimal infrarenal seal and reliable results, even in more challenging ...
-
Manufactured by Mol Scientificbased in CHINA
Mol Scientific offer high-quality Ac-[Cys(Acm)33,42]-EGF (33-42) amide (mouse) product(MPE0012019). Mol Scientific committed to bring our esteemed customers high quality products and the finest services at highly competitive prices. ...
-
Manufactured by Mol Scientificbased in CHINA
Mol Scientific offer high-quality EGF Receptor Substrate 1 product(MPE0011503). Mol Scientific committed to bring our esteemed customers high quality products and the finest services at highly competitive prices. ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you












![Mol Scientific - Model MPE0012019 - Ac-[Cys(Acm)33,42]-EGF (33-42) amide (mouse) Mol Scientific - Model MPE0012019 - Ac-[Cys(Acm)33,42]-EGF (33-42) amide (mouse)](https://d32zuqhgcrpxli.cloudfront.net/ew0KICAiYnVja2V0IjogImVlLWZpbGVzIiwNCiAgImtleSI6ICJmaWxlcy8xMjc0NjEvaW1hZ2VzLzEyNzQ2MV8wXzIwMjIwODE2MDc0NTA5MTY5OTg0NV80MDAucG5nIiwNCiAgImVkaXRzIjogew0KICAgICJyZXNpemUiOiB7DQogICAgICAiZml0IjogImNvbnRhaW4iLA0KICAgICAgImJhY2tncm91bmQiOiB7DQogICAgICAgICJyIjogMjU1LA0KICAgICAgICAiZyI6IDI1NSwNCiAgICAgICAgImIiOiAyNTUsDQogICAgICAgICJhbHBoYSI6IDENCiAgICAgIH0sDQogICAgICAid2lkdGgiOiAyNDAsDQogICAgICAiaGVpZ2h0IjogMTQ1DQogICAgfQ0KICB9DQp9)