Cardiology Catheters Equipment For Medical Equipment
-
Manufactured by Moray Medicalbased in USA
Moray Medical’s initial product is the Coral System, which includes a reusable Driver Assembly, a single-use Clipper toolset, and a Trident input device. The Clipper toolset includes a cardiac-valve-therapy edge-to-edge clip implant that is pre-mounted on a single-use fluid-driven steerable delivery catheter. The Driver, a wireless capital-equipment unit about the size of a brick, ...
-
Manufactured by pfm Medical Agbased in GERMANY
The ASEPT Drainage Kits L allow the out-patient drainage of pleural effusions and ascites. They are available in the sizes 600 ml and 1,000 ml have a low-vacuum which allows a gentle and nearly painless drainage. Apart from that the system can be switched to a gravity application with just one grip. The ASEPT Drainage Kits L in sizes 600 ml and 1,000 ml are equipped with a disk check valve. ...
-
Manufactured by ASAHI INTECC CO., LTD.based in JAPAN
Catheters used for opening up blocked or narrowed blood vessels. Inserted into blood vessels along the PTCA guide wires, it opens up the blood vessels inside by pumping up the balloon with a diameter of 1 to 5 mm placed on ...
-
Manufactured by MICRO-TECH (Nanjing) Co., Ltd,based in CHINA
MICRO-TECH's Disposable Multistage Dilation Balloon Catheter is designed to produce three distinct diameters at three separate pressures during in ...
-
Manufactured by BrosMed Medical Co., Ltd.based in CHINA
With 0.75 mm in diameter, Alveo HP is the smallest high pressure CTO balloon catheter in the world Specifically designed for crossing complex lesions and Chronic Total Occlusions (CTO) as well as tracking tortuous ...
-
Manufactured by Edwards Lifesciences Corporationbased in USA
In the early days of transcatheter mitral valve repair, the goal was a successful procedure that reduced mitral regurgitation (MR) to less than 3+; residual MR grade 2+ was considered acceptable.1,2 Compelling new data on more than 5400 patients show that residual MR grades 0–1+ are significantly associated with superior patient outcomes when compared with residual MR 2+, including ...
-
Manufactured by Merit Medical Systemsbased in USA
The Prelude ACT (Activated Clotting Time) sheaths are part of Merit’s comprehensive family of vascular access products. These half-size sheaths allow easy blood draw through sheath side arm with or without catheters in place. The ACT sheaths accommodate a variety of catheters and other interventional devices to meet your clinical needs. ...
-
Manufactured by GBUK Group Ltd.based in UNITED KINGDOM
Available in straight and right-angled versions with bevelled end for atraumatic insertion. Smooth staggered oval side eyes and open distal tip minimize occlusion and encourage fluid evacuation. Depth markings at 2cm intervals ...
-
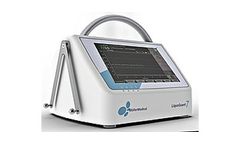
Manufactured by Möller Medical GmbHbased in GERMANY
LiquoGuard is the leading CSF management system that simultaneously measures CSF pressure and drains cerebrospinal fluid under controlled ...
-
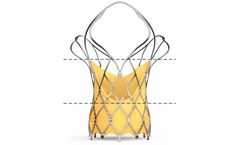
Manufactured by Sahajanand Medical Technologies Ltd.based in INDIA
Self-expanding TAVI device to have 2 rows of marker; First Markers are located at Node 1. Second Markers are located at Node 3. First marker helps; In precise implantation of the valve at the targeted implantation zone. To ascertain the depth of implant. Second marker indicates; When the THV leaflets are going to get ...
-
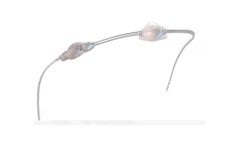
Manufactured by Anuncia Medical, Inc.based in USA
The ReFlow™ System Mini offers the only noninvasive way to attempt to restore, increase, or maintain CSF flow within a shunt system. The ReFlow™ System Mini or the ReFlow™ Mini Flusher may be implanted as part of a shunt system in patients with hydrocephalus or conditions that require draining or shunting the CSF. Without surgery, pressing the ReFlow™ Mini Flusher, which ...
-
by Simeksbased in TURKEY
Simpass NC/HP RX PTCA Balloon Dilatation Catheter performs at great high pressure and non-compliance without compromising any crossability and ...
-
Manufactured by Protembis GmbHbased in GERMANY
ProtEmbo is a catheter-based cerebral protection system to deflect embolic material away from the cerebrovascular circulation during leftsided heart procedures – in particular transcatheter aortic valve replacement ...
-
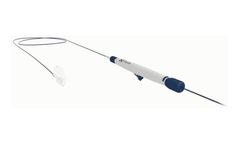
Manufactured by Genodynamic S.R.L.based in ROMANIA
Tip/Shaft French size : 7F. Distal electrode : 4mm. Curve type : B. Color : ...
-
Manufactured by livetec Ingenieurbüro GmbHbased in GERMANY
The RF liveGEN is a specialised high-frequency ablation generator for conducting high-frequency ablation therapies at the human heart or in the renal artery area. To carry out these RF ablation therapies, the RF liveGEN is coupled or integrated into an electrophysiological measuring station system (EPM) with specialised catheters and medical devices and components such as patient monitoring ...
-
Manufactured by NaviGate Cardiac Structures, Inc., (NCSI)based in USA
The GATE™ System is intended to treat functional tricuspid regurgitation (FTR)/tricuspid insufficiency by replacing the function of a patient’s dysfunctional native tricuspid valve with a bio-prosthetic atrioventricular valved stent (AVS). The AVS is designed to restore valve function in patients presenting with FTR/tricuspid insufficiency without the need for conventional, open-heart ...
-
Manufactured by Integer Holdings Corporationbased in USA
We provide a full range of solutions to support every phase of the product life cycle of EP ablation catheters. Design and development of complex ablation catheters including RF ablation catheters and alternative energy ablation catheters (cryo, laser and ultrasound). Transfer of products and processes from one site to another to support product-line transfer, line duplication, low-cost ...
-
Manufactured by MicroPort Scientific Corporationbased in CHINA
Comprehensive solution for the diagnosis and treatment of arrhythmias. FireMagic™ 3D irrigated ablation catheter, combined with Columbus™ 3D EP Navigation System, provides physicians with a comprehensive solution for the diagnosis and treatment of arrhythmias. It helps physicians effectively maneuver the ablation catheter in the body during procedures by providing precise location of ...
-
Manufactured by Osypka AGbased in GERMANY
Connection cables to connect OSYPKA AG ablation catheters with thermistor or thermocouple elements to radio frequency ...
-
Manufactured by Boston Scientificbased in USA
FARAPOINT is a precision focal ablation catheter designed from the ground up with only PFA in mind. In forthcoming clinical studies, FARAPOINT will aim to facilitate highly targeted therapy while leveraging the safety advantages of FARAPULSE PFA. ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you