Cardiac Catheterization (Cardiology Catheters) Equipment
-
Manufactured by Merit Medical Systemsbased in USA
The Prelude ACT (Activated Clotting Time) sheaths are part of Merit’s comprehensive family of vascular access products. These half-size sheaths allow easy blood draw through sheath side arm with or without catheters in place. The ACT sheaths accommodate a variety of catheters and other interventional devices to meet your clinical needs. ...
-
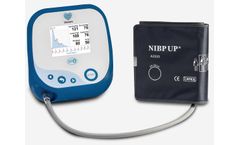
Manufactured by Uscom Ltdbased in AUSTRALIA
The Uscom BP+ is a Supra-systolic oscillometric central blood pressure (cBP) monitoring device which measures blood pressure and blood pressure waveforms at the heart, as well as in the arm; information only previously available using invasive cardiac ...
-
Manufactured by Biocardia, Inc.based in USA
cells at the patient’s bedside. an optimized therapeutic formulation that builds on the total experience in the cardiac stem cell field to-date. a proprietary interventional delivery system that easily navigates a patient’s vasculature to securely deliver the specific dosage of cells in a routine cardiac catheterization ...
-
Manufactured by EKU Elektronik GmbHbased in GERMANY
The maniNO blender for patient supply with a gas blend of oxygen (O2) and nitric oxide (NO) stands out due to easy usability and high NO-precision. It is suited for application in the manual ventilation and can thereby be used in transport as well as in MRI environment. The dosing range of maniNO can be adjusted from 0-40 ppm with an also steplessly adjustable total flow of 0-15 l/min. With a ...
-
Manufactured by Transluminal Technologies, LLCbased in USA
A first-of-its-kind magnesium alloy-based vascular closure device that offers a unique method of quickly achieving stable mechanical vascular closure following percutaneous catheterization for diagnostic or interventional procedures using 5-8 Fr procedural sheaths. The implant consists of a plug and footplate system that when ...
-
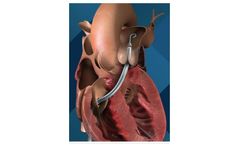
Manufactured by Abiomedbased in USA
The Impella RP heart pump provides temporary, circulatory support for patients who develop right heart failure. It is the only FDA-approved heart pump indicated for patients experiencing acute right heart failure or decompensation following left ventricular assist device implantation, myocardial infraction, heart transplant or open-heart ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you