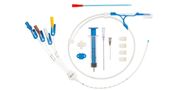
Intravenous Catheters
Articles & Whitepapers
-
Why Sterile Packaging is Crucial for Medical Devices
Patients who need medical treatment often fear being exposed to harmful germs and unsanitary instruments. Just the thought of a healthcare provider using an unsterilized surgical device or needle is enough to make people feel queasy. As a result, patients find it reassuring to watch a clinician remove healthcare devices from sterile packaging before starting a procedure. Sterile packaging ...
-
FDA Approved Peptide Drugs in the First Half of 2023
The FDA's Center for Drug Evaluation and Research (CDER) regulatory approval of a total of 26 NME (New Molecular Entity) drugs in the first half of 2023. It is worth noting that the 26 drugs approved ...
News
-
Bluegrass Vascular Announces Featured Paper in the Journal of Vascular Surgery Reporting on the Use of the Surfacer System
Bluegrass Vascular Technologies, a private medical technology company focused on innovating lifesaving devices and methods for vascular access procedures, announced today the publication of a paper featured on the cover of the June 2022 issue of the ...
Bluegrass Vascular Announces New Paper Reporting Use of the Surfacer System via Transcollateral Approach
FDA grants new QIDP Designation Status for BTX 1801
Shockwave Medical Announces Global Launch of New Peripheral Intravascular Lithotripsy Catheter
Bluegrass Vascular Closes $5 Million Convertible Debt Financing
Equipment & Solutions
-
Showcase
Plaster with Hard-Wearing Hook and Loop Straps
Main-Lock is a skin-friendly plaster with hard-wearing hook and loop straps which is used i.a. for the securement of infusion tubing systems, arterial or central venous catheters and feeding tubes.