Medical Device Testing News
-
Protheragen-ING Lab, a Trustworthy GLP Service Provider, Unveils Its Cutting-Edge Medical Device Services
Protheragen-ING Lab, a well-known and reputable Good Laboratory Practices (GLP) service provider, has recently unveiled its cutting-edge medical device services. With a track record of excellence and reliability in providing high-quality services to the pharmaceutical and biotechnology industries, Protheragen-ING Lab is now expanding its offerings to include comprehensive solutions for medical ...
-
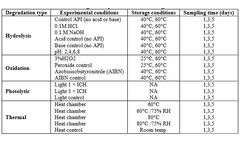
STEMart Launches New Force Degradation Services to Ensure Drug Stability and Safety
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and efficacy throughout their shelf life. Forced ...
By STEMart
-
STEMart Launches Biological Indicator Sterility Testing Service for Medical Devices
STEMart, a U.S.-based provider of comprehensive services for all stages of medical device development, has recently introduced Biological Indicator Sterility Testing services for the industry of medical devices. This testing is performed according to the exposure of Biological Indicators (BIs) after completion of the sterilization load, and it is a qualitative test to reveal results that can ...
By STEMart
-
Terumo India Signs a New Strategic Commercial Distribution Partnership with Sensible Medical Innovations
Introduces medical radar monitoring and imaging technology (ReDS™) to Indian patients for the first time Addresses an unmet medical need for over 8 million patients across the country Terumo India, the Indian arm of Terumo Corporation (TSE: 4543), a global leader in medical technology, today announced that it has signed a partnership with Sensible Medical Innovations, the market ...
-
Nalu Medical, Inc. Names Thomas West President and Chief Executive Officer
Nalu Medical, Inc. (“Nalu”), a private company focused on innovative and minimally invasive solutions for chronic neuropathic pain, announced today that the Board of Directors has appointed Thomas “Tom” West as President and Chief Executive Officer. This leadership appointment is effective on August 8th and follows the decision by Earl Fender, Nalu’s current CEO, to ...
-
STEMart Launches Microbiology & Sterility Testing Services for Medical Devices
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, introduces comprehensive microbiology and sterility testing for sterile, non-pyrogenic products. STEMart has extensive expertise in microbiology as well as sterility testing and remains dedicated to a full-service experience that supports manufacturers in meeting ...
By STEMart
-
STEMart Introduces Biocompatibility Evaluation of Breathing Gas Pathways in Medical Devices
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, expands its Biocompatibility Testing services and introduces biocompatibility evaluation of breathing gas pathways in medical devices. This new testing follows the biocompatibility guidelines modified for medical devices and uses a solid scientific rationale. ...
By STEMart
-
STEMart Launches Pyrogenicity Testing Service for Medical Devices
STEMart, a provider of integrated medical device CRO services dedicated to medical device and diagnostic clinical development, now launches Pyrogenicity Testing service for the medical device industry. This new testing follows the biocompatibility guidelines modified for medical devices. Due to the risks associated with medical devices, comprehensive medical device testing throughout the product ...
By STEMart
-
Nalu Medical Releases “Haleakala” Update, Extending Pulse Width Range and Unlocking the System’s Patient Activity Monitor
Nalu Medical (“NALU”) announces the “Haleakala” update, substantially upgrading both the software and firmware of their award-winning Nalu Neurostimulation System. This update adds significant new capabilities for both spinal cord stimulation (SCS) and peripheral nerve stimulation (PNS). The update broadens the stimulation pulse width parameter to the widest range ...
-
Formal System Testing for Medical Devices – Part 2
Verification is critical for medical device testing, which is why we recommend reading part one if you haven’t already. In part one of this series, we examined the need for medical testing and briefly covered the first few aspects of device verification. In this article, we’ll cover the verification dry run and the formal verification process that makes new medical products a reality. ...
-
Nalu Medical Named Best MedTech Company and Medical Device in Field of Neurology
Nalu Medical is pleased to announce its flagship micro-Implantable Pulse Generator (mIPG™) was selected as the 2021 winner of the MedTech Visionaries Award for Best MedTech Company and Best Medical Device in the field of neurology. The MedTech Visionaries Awards honor organizations that apply science and technology to real-world problems to advance MedTech solutions for businesses and ...
-
Nalu Medical kicks-off ‘Engineered to be More’ with new product releases and software upgrade to provide more therapy options and support for its patients and physicians
Nalu Medical, Inc. (“Nalu”), a Carlsbad, California-based company that has successfully miniaturized neurostimulation implants for the management of intractable chronic pain for both Spinal Cord Stimulation (SCS) and Peripheral Nerve Stimulation (PNS) indications, launches its ‘Engineered to be More’ branding campaign. The campaign highlights the award-winning engineering ...
-
Neuspera Medical Completes First Implants of its Neuromodulation Device in its two-stage Pivotal Clinical Trial to treat Urinary Urgency Incontinence
Neuspera Medical Inc., a clinical stage, private venture capital backed neuromodulation company, today announced the closing of the company’s $65 million series C equity financing. The Series C round was co-led by Vertex Ventures HC and Treo Ventures. Lori Hu, a Managing Director at Vertex Ventures HC and Mudit K. Jain, Ph.D. Managing Partner, Treo Ventures, joined the Board of Directors. ...
-
Guided Therapeutics Receives News from Chinese State Food and Drug Administration that LuViva Meets Product Technical Requirements
Guided Therapeutics, Inc. (OTCQB: GTHP), the maker of the LuViva™ Advanced Cervical Scan, based on its patented biophotonic technology, announced today its Chinese co-manufacturing partner and distributor for China, Shandong Yaohua Medical Instrument Corporation (SMI), has received notice from the Jinan Medical Device Quality Supervision and Testing Center of the State Food and Drug ...
-
US. Sen. Tammy Baldwin visits Attwill Medical Solutions
Attwill Medical Solutions was proud to host U.S. Senator Tammy Baldwin (D-WI) today for a. roundtable discussion regarding the company, as well as a comprehensive factory tour. Attwill Medical. Solutions is one of the largest manufacturers of reagents for COVID-19 and other PCR testing kits, and. has unique freeze-drying technology, which may be utilized for the development of vaccines in a pill. ...
-
Nalu Micro-Implantable Pulse Generator Receives Award for Implanted Medical Device Innovation and Health Benefits
Nalu Medical, Inc. (“Nalu”), a California-based company that has successfully miniaturized neurostimulation implants for chronic intractable pain management indications, announces its Nalu micro-Implantable Pulse Generator (mIPG) was named the Implant and Tissue-Replacement device Gold Winner at the 2021 Medical Design Excellence Awards (MDEA). The MDEA is a prestigious award that ...
-
Nalu Medical Releases Latest Software Upgrade to Expand Nalu Neurostimulation System Capabilities
Nalu Medical, Inc. (“Nalu”), a Carlsbad, California-based company that has successfully miniaturized neurostimulation implants for the treatment of intractable chronic pain via Spinal Cord Stimulation (SCS) and Peripheral Nerve Stimulation (PNS), announced the release of their latest software upgrade that adds new capabilities to their micro neurostimulation system. Like a smartphone ...
-
Nalu Micro-Implantable Pulse Generator Receives Expanded FDA Label for Service Life of 18 Years
Nalu Medical, Inc. (“Nalu”), a California-based company that has successfully miniaturized neurostimulation implants for the treatment of chronic pain via Spinal Cord Stimulation (SCS) and Peripheral Nerve Stimulation (PNS), in July 2020 received clearance from the U.S. Food and Drug Administration (FDA) of an expanded label for its micro-Implantable Pulse Generator (mIPG) to an ...
-
Premier Inc. Awards BioSerenity Contract
BioSerenity, Inc., a leading global provider of diagnostic solutions in the areas of Sleep Medicine, Neurology, and Cardiology, announced today it has been awarded a group purchasing agreement for PP-SV-268 by Premier as part of its Preferred Partner agreement. The prevalence of sleep disorders, epilepsy and cardiovascular disease pose a significant challenge to the US healthcare system. ...
By BioSerenity
-
BlueWind Initiates U.S. Enrollment in Clinical Trial of the RENOVA iStim to Treat Overactive Bladder
BlueWind Medical announced today that enrollment has commenced at U. S. sites in the Pivotal Clinical Trial of the RENOVA iStim™ implantable tibial neuromodulation System (RENOVA) for the treatment of Overactive Bladder (OAB) following conditional Investigative Device Exemption (IDE) approval by the U.S. Food and Drug Administration (FDA). There are approximately 40 million adults in the ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you