Medical Devices News
-
Showcase
Mojo Vision Reaches Significant Development Milestone for Smart Contact Lens
New prototype of Mojo Lens incorporates numerous industry-first features, including the world’s smallest and densest dynamic display, low-latency communication, and an eye-controlled user interface SARATOGA, Calif.--(March 30, 2022)--Mojo Vision, the Invisible Computing Company, today announced its most advanced prototype of Mojo Lens, the world's first true smart contact lens, including ...
-
Showcase
Koning Ends the Year with $2.6M Republic Raise
NORCROSS, Ga., Feb. 1, 2022 /PRNewswire/ -- Koning, a growing medical device startup company based outside Atlanta, had a successful conclusion to 2021. The company recently finished their first Republic crowdfunding campaign, exceeding capital targets, and raising over $2.6M in only 5 months. Koning's FDA breast screening trial is also underway with participating clinics in Daytona Beach, FL and ...
-
Showcase
Neeuro (Singapore) and epihunter (Belgium) partner to create new and innovative digital solutions for people with brain disorders
On March 15th 2015, Tim Buckinx’ son said: “Papa, you work in digital, can’t you create a light that turns on when my brain switches off?” At ten years old, he was simply fed up with the daily impact of his refractory epilepsy. That pain became a vision, and that vision became the Belgian digital therapeutics company epihunter, aimed at enabling people with a brain ...
By Epihunter NV
-
Showcase
Atmo Biosciences awarded Victorian Government Technology Adoption and Innovation Program grant to support software development
Atmo Biosciences has been awarded a Victorian Government Technology Adoption and Innovation Program (TAIP) grant to support the software development for the Atmo Gas Capsule. The TAIP supports Victorian small to medium size enterprises (SMEs) to adopt or develop innovative, new and commercial technology. Atmo was awarded the full $50,000 grant amount, which will go towards hiring a Principal ...
-
Showcase
Corify Care raises €2.35M to translate its cardiac mapping technology to the market. .
- Corify Care is developing ACORYS®, a non-invasive cardiac mapping medical device to transform the cardiac arrhythmia treatment process. - The investment round is led by Clave Capital, counting with investment from CDTI through its Innvierte program and the support from Genesis Biomed. Corify Care, the developer of an innovative and completely non-invasive cardiac mapping system, ...
-
Showcase
Neurescue`s Breakthrough Intelligent Balloon Catheter FDA 510(k) Cleared for Hemorrhage Control and IDE Approved for Cardiac Arrest
COPENHAGEN, Denmark--(BUSINESS WIRE)--Neurescue, a medical device company developing innovative cardiovascular solutions to improve the outcomes for emergency patients, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to market the company’s NEURESCUE® device for temporary occlusion of large vessels, including patients requiring emergency ...
By Neurescue
-
Showcase
Corify Care participated in the CEI Satellite Meeting CinC 2020: Challenges in the Daily Application of ECGI
Last 18th of September, Corify Care CEO and co-founder, Dr. Andreu Climent participated in the CEI Satellite Meeting CinC 2020: ECGI Past, Present, and Future: from Engineering to Clinical Application organised by the Consortium for ECG Imaging. The objective was to discuss engineering hurdles for ECGI being incorporated into clinical practice and are partnering with industry leaders to ...
-
Showcase
Empatica partnering with U.S. Army MRDC to deploy COVID-19 early detection wearable
BOSTON, Nov. 6, 2020 /PRNewswire/ -- Empatica has been selected by the U.S. Army Medical Research and Development Command (USAMRDC) to deploy a wearable and algorithm that enable the early and pre-symptomatic detection of COVID-19. The project's focus is on protecting the health of the general population by preventing outbreaks via early detection and alerting. Empatica's Aura algorithm gives a ...
-
Showcase
Empatica and BARDA Join Forces to Validate Wearable System That Detects COVID-19 Before Symptoms Appear
BOSTON, June 19, 2020 /PRNewswire/ -- Digital healthcare company Empatica announces a pioneering new partnership with the Biomedical Advanced Research and Development Authority ("BARDA"), part of the Office of the Assistant Secretary for Preparedness and Response at the U.S. Department of Health and Human Services (HHS), to validate an early warning system for COVID-19 and other respiratory ...
-
Showcase
Zeneo® Adrenaline: A New Innovative Formulation
Anaphylactic shock is an absolute medical emergency. When anaphylactic shock is suspected, emergency services must be called and emergency first aid administered immediately. The only first-line treatment for anaphylactic shock is injectable adrenaline1. In the USA: 1.6% to 5.1% of the population has experienced anaphylactic shock, 1% of hospitalizations are caused by it, 0.1% of patients ...
By Crossject
-
STEMart Announces Pilot Production Services for Medical Devices
STEMart, a US-based provider of comprehensive services for all phases of medical device development, is proud to announce the launch of its new Pilot Production Services for the medical device industry. These services allow manufacturers to test and refine their products prior to full-scale production, streamlining the development process of new medical devices and minimizing risk to save time ...
By STEMart
-
Infection Control Resources to Identify Klebsiella and Other Pathogens in Healthcare Settings throughout Puerto Rico
The Centers for Disease Control and Prevention (CDC) describes Klebsiella as a type of gram-negative bacteria that can cause different types of healthcare-associated infections (HAIs). These include pneumonia, bloodstream infections, wound or surgical site infections, and meningitis. The agency warns Klebsiella bacteria have increasingly developed antimicrobial resistance, most recently to the ...
-
Infection Control Resources to Mitigate Patient Exposure Risks to Klebsiella and Other Pathogens
An article published last year in Emerging Infectious Diseases detailed a notable increase of carbapenem-resistant Klebsiella pneumoniae (CRKP) infections in New York City during 2021 through June of 2022. The data for the study came from a large public healthcare system in New York City that serves more than 1.2 million people each year. The Centers for Disease Control and Prevention (CDC) ...
-
Alfa Chemistry Recently Launched a Sub-website for Functional Polymers
The US-based chemical supplier Alfa Chemistry announced the launch of a new sub-website for the supply of functional polymers, including Adsorptive Polymers, Biomedical Polymers, Electrofunctional Polymers, Functional PEGs, Photoactive Polymers, Silicone Polymers and more. With unstopping efforts, the company continues to offer chemicals for specialized applications, such as catalysts, fatty ...
-
Klebsiella in Healthcare Settings Discussed in New Online Video
The IAQ Video Network and Cochrane & Associates announced the release of their latest educational video today. Their newest production discusses Klebsiella, a type of gram-negative bacteria that can cause various healthcare-associated infections (HAIs). “HAIs due to Klebsiella can include pneumonia, bloodstream infections, wound or surgical site infections, and meningitis,” said ...
-
EMI/RFI: Materials and Technologies - A Comprehensive Market Analysis by BCC Research
Boston: “According to the latest BCC Research study, the demand for EMI/RFI: Materials and Technologies is estimated to increase from $7.4 billion in 2023 to reach $9.2 billion by 2028, at a compound annual growth rate (CAGR) of 4.6% from 2023 through 2028.” This comprehensive report delves into the dynamic landscape of the EMI/RFI materials and technologies market, meticulously ...
By BCC Research
-
Caresyntax Announces Highlights of 2023
Caresyntax, the leading vendor of enterprise-scale data-driven surgical intelligence solutions to make surgery safer and smarter, today announced its highlights and achievements of 2023, as the company has continued its strong growth journey. Dennis Kogan, co-founder, and CEO: “Caresyntax celebrated its 10-year anniversary in 2023 and accelerated our journey of innovation and expansion ...
By Caresyntax
-
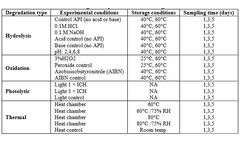
STEMart Launches New Force Degradation Services to Ensure Drug Stability and Safety
STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and efficacy throughout their shelf life. Forced ...
By STEMart
-
Rescale and Caresyntax Expand their Collaboration to Provide Scalable Platform for AI-Powered Simulation and Intelligence in Surgery. World Economic Forum Collaboration Enhances Access to Surgical Care and Contributes to Health Equity
Rescale, the leader in cloud high performance computing (HPC) to accelerate innovation and research, and Caresyntax, the leading vendor-neutral surgical data and intelligence platform converging AI-powered software, devices, and clinical services, announced a broader collaboration through the World Economic Forum Open Forum for AI in Health and Healthcare Communities. This collaboration marks ...
By Caresyntax
-
Optimizing Monoclonal Antibody Production: Temperature And Osmolality’s Impact, Upcoming Webinar Hosted By Xtalks
In this free webinar sponsored by Advanced Instruments, gain insights into advances in monoclonal antibody production technology and manufacturing strategies. Attendees will learn why temperature and osmolality in cell culture are critical parameters affecting cell growth, viability and the concentration of HCPs in the supernatant. The featured speaker will discuss how temperature and osmolality ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you