Medical Devices Software For Medical Equipment
-
Manufactured by Cisa Group S.R.L.based in ITALY
TraceCare® system offers a number of packages depending on the customer’s needs. MONITORING: The Monitoring version allows the monitoring and saving of the cycles of the machines present within the CSSD. Data concerning the main cycle parameters are saved in the DataBase and can be displayed as graphs or shown in summary reports. The communication between TraceCare® and the PLC ...
-
by Epihunter NVbased in BELGIUM
Epihunter Pro makes the automatically built seizure diary available through a webportal for the physician. The epihunter home-care solution is based on a wearable EEG headband and a smartphone with a detection algorithm. When a seizure is detected, the people around are notified immediately. Next to this, an entry is automatically logged in the seizure diary. Also, a video can be automatically ...
-
by Qynapsebased in FRANCE
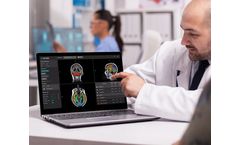
QyScore is a groundbreaking neuroimaging analysis platform for both clinical routine and clinical trial settings. One scan at a time, QyScore has the potential to revolutionize the treatment of CNS disease and create the potential for earlier precision on the frontlines of CNS disease. QyScore is intended for automatic labeling, visualization and volumetric quantification of segmentable brain ...
-
based in USA
The only seizure monitoring, alerting, and reporting application that does it all. We chose to embed the Overwatch App into the Apple® Watch to ensure reliability, accessibility, and convenience. From real-time notifications to collecting data in an online, daily seizure log, the Overwatch App works hard to make living with epilepsy safer and more manageable. ...
-
based in ISRAEL
Adhere to strict MDR/FDA regulations and ISO quality standards by eliminating paper-based QMS ...
-
by Enhatch Inc.based in USA
Hospitals around the country are limiting the number of people permitted in operating rooms. Infection control and security were the main reasons why entry into the operating room is much tighter. RepVision is a HIPAA compliant surgical application designed for medical device companies to remote into a ...
-
Manufactured by Micrel Medical Devices SAbased in GREECE
Rythmic Connect is a wireless real time technology using a GPRS device (IP Connect Pack) that gets connected to the ambulatory Rythmic™pump and enables the pump to communicate with a web server and provide its infusion status online through the MicrelCare system. Physicians, nurses and homecare service providers can access the status of their patients' infusion online from anywhere. ...
-
Manufactured by Oncomfortbased in BELGIUM
Digital Sedation™ is a safe and effective digital therapy to manage patients’ pain and anxiety before, during and after medical procedures, without medication. CE-marked software as a medical device; The application has been co-created with multidisciplinary medical experts having deep clinical and scientific knowledge in this field. The Digital Sedation™ is tailored to the ...
-
based in CANADA
100% software solution that integrates vital signs information from medical devices with the patient record. Enovacom Patient Connect is a unique connectivity solution that extracts vital signs and other patient information from biomedical devices and integrates this directly with the patient record. This 100% software-based solution delivers integrated vital signs information via a single ...
-
based in ISRAEL
Our medical requirements management tool is perfect for manufacturers of medical devices and will provide you with a number of essential quality-related ...
-
by Centaur Labsbased in USA
Audio clips from medical devices are critical indicators of patient health, and can be leveraged to train predictive algorithms that detect heart conditions, lung abnormalities, and more. Our annotation platform is tailored specifically to medical audio clips, with features such as range selection, spectrogram integration, and motion data ...
-
Manufactured by AioCarebased in POLAND
Limited access to specialist doctors and to the appropriate medical equipment means that nearly 2/3 of patients are incorrectly diagnosed at the first symptoms of pulmonary ...
-
based in BELGIUM
FDA 510(k)-cleared D2P® software and 3D Systems printers allow surgeons, radiologists, lab technicians, and device designers to create diagnostic-quality digital 3D models and physical 3D prints easily and ...
-
Manufactured by Caresyntaxbased in GERMANY
Educate and improve your surgical teams with data-driven training and effective surgical performance ...
-
by Centaur Labsbased in USA
Waveforms are critical indicators of patient health, and can be leveraged to train predictive algorithms that detect heart, lung, neurological conditions, and more. Our annotation platform enables clients to structure waveforms by both classifying features within the waveform and segmenting regions of ...
-
by Vintarabased in USA
Vintara’s Enterprise Portal helps leaders deploy world class best practices for the manufacture and sale of medical devices. Our configurable workflow applications will drive performance and world class regulatory compliance including: ISO 13485, 21 CFR Part 11 compliance, 21 CFR Part 820 compliance, CAPA, Device Master Records, Customer Complaints, ...
-
Manufactured by Voyager Imaging a division of Intellirad Solutions Pty Ltdbased in AUSTRALIA
Voyager PACS is a web enabled, server based PACS that provides medical images of any patient to any doctor, anytime, anywhere. Voyager PACS provides image archiving, soft copy reporting, management and distribution functionality. The system receives images from modalities or from other sources (i.e. Teleradiology) via DICOM and stores these on the PACS Server or attached storage device. The ...
-
Manufactured by Shoulder Innovationsbased in USA
Genesis is an early stage investor in medical device technology. We are differentiated through our deep level of engagement in delivering value through all stages of the commercialization process. This is accomplished by leveraging the time, talent, and financial resources of the Genesis Innovation Group Portfolio. ...
-
based in USA
OOS Software to Automate Out of Specification (OOS) Processes to Comply with FDA CGMPs and 21 CFR Part 211 and Part 820 Regulations. In the FDA environment, specifications are essential in maintaining quality. CGMP regulations for finished pharmaceuticals (21 CFR Parts 210-211) and medical devices (21 CFR Part 820) require strict conformance to approved specifications which can be acheived easily ...
-
based in USA
Variant Interpretation * Variant Tracking * Coverage Confirmation Save Time & Resources * Compatible with data from all NGS systems * Targeted Panels and Whole Exome Sequencing. Developed in collaboration with the Laboratory Medicine, Information Technology, and Health Science Research departments of Mayo Clinic, Geneticist Assistant NGS Interpretative Workbench is a unique tool for the ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you