Medical Devices Training For Medical Equipment
-
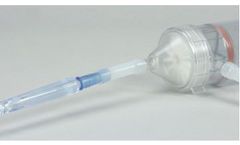
Manufactured by Sisu Global Healthbased in USA
During this training you will gather knowledge about Heamfuse as a product as well as been given the tools for Sales Learning and Common Objections in the medical device Sales Field. ...
-
based in USA
This comprehensive training package of four courses covers clinical trial monitoring techniques, audit procedures and the setting-up of Data Monitoring Committees ...
-
Manufactured by Proxomed Medizintechnik GmbHbased in GERMANY
Our training department focuses strongly on how therapeutic training is deployed. To allow faster and more efficient use of the devices, you will receive user training for the hardware and software of our therapeutic and training ...
-
based in USA
Emergencies can be terrifying, and sometimes, those who witness them have to respond right on the spot.Emergencies happen in the blink of an eye. To save lives, any witness must be as swift to react. This program illustrates what to do if a person meets a life-threatening medical emergency. It includes correct procedures for dealing with: Heart attacksChoking StrokeAsthmaDiabetesSeizure and ...
-
based in SOUTH KOREA
KTR operates a separate website (www.ktr.or.kr/edu) that provides education/training related tasks including introduction of training programs, applications for training, and education/training history ...
-
based in USA
Although the ISO 9001 requirements have been removed from IATF 16949:2016 and are published as a separate ISO standard, IATF 16949 is not a stand-alone standard and must be used in conjunction with ISO 9001, we offer courses that cover both the ISO 9001:2015 and IATF 16949:2016 requirements. Group exercises and case studies with examples from the automotive industry will be used to develop the ...
-
based in USA
This medical device software development, verification and validation training package of four courses covers wellIEC 62304 requirements and explains risk based approach for validation using validation protocols and GAMP®. Medical device software can make or break a device. Badly developed, non-compliant software can compromise a device's safety and reliability. These four webinar recording ...
-
Manufactured by Nexus CMFbased in USA
This is a weekend, hands-on cadaveric training course demonstrating the McCain Technique and utilizing the McCain TMJ Arthroscopy System and the Nexus CMF ClearView Hybrid Tower. Attendees are encouraged to bring surgical staff at no additional charge. The training is structured to provide attendees with hands-on cadaveric exposure designed to increase the participant’s ability to perform ...
-
by Marcus Evansbased in UNITED KINGDOM
Mastering all aspects of present-day marketing through highly practical workshops with real life examples and discussions with like-minded professionals. This series includes spin-off programmes tailored to help maintain a competitive edge whilst boosting success in the market. Selection of courses relevant for all marketing practitioners’ levels focusing on Marketing, Marketing ...
-
based in USA
This comprehensive training package of 8 courses is aimed at helping medical device companies ensure that their supply chain controls and manufacturing processes comply with existing ...
-
based in USA
The main purpose of stability program is to evaluate a shelf life for a product to enter a market and before that determine a safe stability profile during clinical trial. There are many guidance which showcase the best practices how to achieve a successful stability program and then specific guidelines are formulated for a particular product ex – ICH regulatory guidance, QBD, PQS, ...
-
based in USA
The following is a pack of 8 best-selling training CDs for regulatory professionals in the Medical Device industry. These courses provide information on how medical device companies can achieve compliance in the important stages of device development, marketing and ...
-
based in USA
Many regulated companies have not brought their old products up to current FDA standards and are not in total compliance. This webinar defines the criteria to be used for medical products design history file remediation. For example companies should be compliant to 14971:2007. Design History Files need to be updated to meet current standards. The FDA expects risk management to be integrated into ...
-
Manufactured by TSK Groupbased in CANADA
TSK aesthetic training partners offer aesthetic courses for all experience levels. TSK Laboratory offers you a unique opportunity to attend a highly-specialised medical aesthetic masterclasses led by multi-award-winning tutors and key opinion leaders. In these aesthetic training courses, elite tutors will share their in-depth knowledge and experience through one to one guidance and detailed ...
-
Manufactured by NovaDigm Therapeutics, Inc.based in USA
Staphylococcus aureus (S. aureus) is the leading cause of skin and soft tissue infections (SSTI), also referred to as skin and skin structure infections. Methicillin-resistant S. aureus (MRSA) is a predominant cause of hospital- and community-acquired SSTI. Recent studies conducted by the US Department of Defense Tricare health system have estimated rates of S. aureus SSTIs (54% MRSA) among their ...
-
based in USA
This presentation will review a methodology for implementing Risk-Based Validation of test equipment, facilities and analytical instruments used in GLP, cGMP and GCP facilities. A case study is presented for the construction qualification, relocation and new equipment installation for a cGMP facility. The cost and time savings associated with using risk-based validation will be presented. Over ...
-
Manufactured by Sisu Global Healthbased in USA
This course will allow learners to understand the operation and use case for the HEMAFUSE ...
-
based in USA
ComplianceOnline has created these courses individually through world renowned experts and combined them together to create a comprehensive training resource for companies to train their employees. You can store it as a library material and train all your employees in these compliance issues to get them ready to face any kind of compliance challenge and overcome it through expert advice and best ...
-
based in USA
The following is a pack of 5 best-selling training CDs for regulatory professionals in the Medical Device industry. These courses provide information on how medical device companies can achieve compliance in the important stages of device development, marketing and maintenance. In today's complex regulatory environment, medical device manufacturers often find that the road to compliance can be ...
-
based in UNITED KINGDOM
SES run regular scheduled 3 day First Aid at Work training courses from our training centre in Essex. We can also run 'on site' First Aid at Work courses at your premises or arrange a course specifically for you at our training centre. All of our First Aid Courses are regulated by ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you