Medical Systems Software For Medical Equipment
-
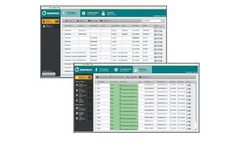
Manufactured by Medset Medizintechnik GmbHbased in GERMANY
HIS-Integration, Connection with existing medical systems, Integration with ECG-Writer, ...
-
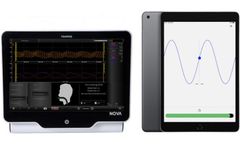
Manufactured by Finapres Medical Systemsbased in NETHERLANDS
New GAT application enables standardization and quantification! In autonomic testing, standardized procedures are essential for reliable test results. Finapres Medical Systems developed a Guided Autonomic Testing (GAT) application as part of the Finapres® NOVA, which guides the operator and the patient through a series of autonomic test ...
-
based in USA
A leading differential diagnosis tool relied upon by thousands of clinicians every ...
-
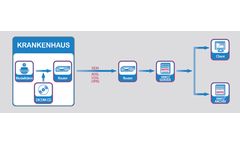
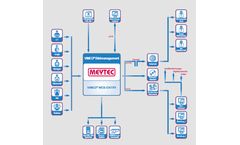
Manufactured by Meytec GmbHbased in GERMANY
Effective image communication via DICOM-network protocol. The telemedicine solution VIMED®TRANSFER enables the direct and inter-divisional image communication via DICOM-network protocol between a workstation and a remote VIMED SERVER. VIMED TRANSFER supports the transmission of the extensive image data via fixed and mobile transmission channels with the use of the lossless compression ...
-
based in UNITED KINGDOM
30,000 Evidence-Based Clinical Decision Support Tools, Support diagnostic results and document medical necessity, Automate medical guidelines and integrate with EHRs, Use analytics in medical teaching and seminar ...
-
Manufactured by Finapres Medical Systemsbased in NETHERLANDS
New GAT application enables standardization and quantification! In autonomic testing, standardized procedures are essential for reliable test results. Finapres Medical Systems developed a Guided Autonomic Testing (GAT) application as part of the Finapres® NOVA, which guides the operator and the patient through a series of autonomic test maneuvers. The GAT application consists of a graphical ...
-
Manufactured by Pleora Technologies Inc.based in CANADA
Streamline end-to-end data delivery and application development for mission-critical automation, security & defense, and medical ...
-
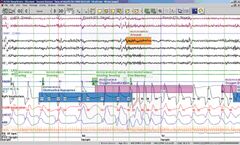
Manufactured by Natus Medical Incorporated (Natus)based in USA
Natus SleepWorks is an extensive, flexible and cybersecure sleep diagnostic solution that simplifies the setup, collection, review, analysis and reporting of comprehensive sleep studies. Efficiency is optimized using HL7 interfaces for bi-directional data exchange with electronic medical record systems. Video and PSG data can be analyzed and reviewed while the study is being collected. Customized ...
-
Manufactured by DYSIS Medical Ltd.based in USA
DYSIS View is an out of the box ready, compact and portable colposcope with computer-aided cervical mapping that generates the data to help healthcare professionals detect cervical lesions efficiently and direct the patient ...
-
Manufactured by Codonicsbased in USA
Codonics award-winning Safe Label System (SLS) is the world’s leading solution for safe, compliant, and fast medication labeling — anywhere medications are prepared! An FDA Class 2 medication safety system, SLS uses barcode technology to improve safety, compliance and efficiency during medication preparation. SLS helps to eliminate vial and ampoule swaps, mislabeling, and syringe ...
-
Manufactured by Meytec GmbHbased in GERMANY
Allows fast and secure transmission, visualization and findings of the medical images via networks in intranet or internet. Advanced and cost-effective teleradiology solution for radiological hospitals and practices. Important extra module for finding and viewing of neuroradiological DICOM-images by the telemedical collaboration of radiology, stroke unit and tele-stroke unit. ...
-
based in ISRAEL
The software for running mammography centers will allow you to enjoy a variety of advantages and tools that can assist you in significantly streamlining and improving the institute’s work processes. The MEDSOFT PRO Mammo software for running mammography centers provides the best answers to the needs of managing the institute and is completely configured to this specific ...
-
Manufactured by LM Medical Division s.r.l.based in ITALY
Benefits for Clinical Staff: Thanks to the Control Panel the medical team can receive information about the medical gas system, HVAC plant and electrican power supply system. Design: Attractive, enticing and modern solution and graphics not only make the CP easy to use but also complement the pleasing appearance of today’s high tech operating ...
-
based in INDIA
Birlamedisoft presents Medical College Management System with complete student and education management ...
-
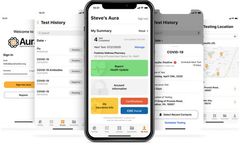
based in USA
GENETWORx Laboratory’s unique software platform, Aura, provides employers with an easy tool for scheduling, receiving, and managing test results. ...
-
based in CANADA
100% software solution that integrates vital signs information from medical devices with the patient record. Enovacom Patient Connect is a unique connectivity solution that extracts vital signs and other patient information from biomedical devices and integrates this directly with the patient record. This 100% software-based solution delivers integrated vital signs information via a single ...
-
based in GERMANY
MediConnect® is a modern, flexible, future-proof system that organizes processes for planning, presenting, communicating, and archiving patient data. This standardization provides work flows with efficiency and simplicity. The programming architecture of MediConnect® is based on the Microsoft .NET Framework© in combination with the Microsoft SQL Server©. This unique ...
-
based in GERMANY
Medication processes integrated into hospital information systems are supported by the ID MEDICS® expert software from ID, the market leader in software for revenue-optimized coding and safe medication. ID MEDICS® has been designed as a specialist application for medication, to complement hospital information systems. As a medication backbone, it provides uniformly prepared medication ...
-
Manufactured by Lite-Med Incbased in TAIWAN
Technology.Human Nature. Chinese-English bilingual interface. Easy to use. Support multiple stones. Report by month, quarter, annual or customized the period can be exported. Customizable report (optional; with additional ...
-
based in USA
The medical device reporting (MDR) requirements of 21 CFR 803 apply to manufacturers, importers, distributers, and device user facilities in the United States. Such facilities are required to report adverse device-related events to the Food and Drug Administration (FDA) or the device manufacturer, and occasionally to both. These requirements are supplemental to the quality system requirements of ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you