Prosthesis News
-
Showcase
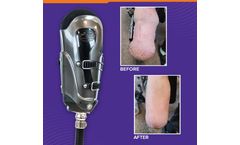
The iFIT device gently and comfortably reshapes the residual limb.
This patient reduced their volume within a month period of time using the iFIT transtibial prosthesis. The iFIT system relieves swelling and shapes the limb quickly. There is no need to wait for prosthetic shrinker socks to reduce volume, so those with limb loss can be fit right away and get back on their feet. ...
-
Photodisinfection successfully eradicates pathogens associated with replacement joint infections
A research team in Zurich, Switzerland has presented results from an in vitro study showing that photodisinfection achieved >99.99% eradication (4-7 log10 kill) of the key pathogens in biofilms associated with implantable joint infections, with no reported regrowth. These results were presented at the recent Swiss Society for Microbiology (SSM) Annual Congress 2022 in Lausanne, Switzerland. ...
-
enVVeno Medical Reports Second Quarter 2022 Financial Results and Provides Corporate Update
Ended the quarter with $47.2 million in cash and investments – expected to fund operations through the end of 2024, including release of topline SAVVE pivotal trial data IRVINE, CA / August 3, 2022 / enVVeno Medical Corporation (NASDAQ:NVNO) (“enVVeno” or the “Company”), a company setting new standards of care for the treatment of venous disease, today provided a ...
-
reSept ASD Occluder implanted in EU for the first time
French TV France3 reported on the first patients implanted with a reSept ASD Occluder in France and in the European Union at Bordeaux University Hospital. The coverage includes video interviews with young patient Léo and his mother, and with the doctors that treated him, as part of the ASCENT ASD trial. Professor Jean-Benoît Thambo, head of the department of pediatric and congenital ...
-
enVVeno Medical Reports First Quarter 2022 Financial Results and Provides Corporate Update
Ended the quarter with $51.3 million in cash – expected to fund operations through the end of 2024 including release of topline pivotal trial data Ongoing advancement of SAVVE U.S pivotal trial with 20 sites available to enroll patients Progress on development of second device for the treatment of venous disease expected to be unveiled in mid-2022 IRVINE, CA / May 2, 2022 / enVVeno ...
-
enVVeno Medical Reports Fourth Quarter and Year-End 2021 Financial Results and Provides Corporate Update
Ended the Year With $55 Million in Cash Expected To Fund Operations Through the End of 2024 Including Release of Topline Pivotal Trial Data Nine (9) VenoValve Surgeries Successfully Completed and 16 Clinical Sites Currently Active in Ongoing SAVVE U.S Pivotal Trial Continued Advancement in Development of Second Device for the Treatment of Venous Disease Expected To Be Unveiled in Mid-2022 ...
-
Lineage Announces Pipeline Expansion to Include Auditory Neuronal Cell Therapy for Treatment of Hearing Loss
Expansion of Pipeline Into a Third Neuronal Cell Type Builds on Existing Capabilities Intellectual Property Has Been Filed Covering Composition and Methods for Generating Auditory Neuronal Progenitors Hearing Loss Afflicts More Than 5% of the Population; More Than 430 Million People Lineage Cell Therapeutics, Inc. (NYSE American and TASE: LCTX), a clinical-stage biotechnology company ...
-
Impact of Time to Receipt of Prosthesis on Total Healthcare Costs 12 Months Postamputation
Earlier receipt of a prosthesis is associated with reduced spending in the 12 months post-amputation of approximately $25,000 compared with not receiving a prosthesis. The results of this study suggest that not providing or delaying the provision of a prosthesis increases costs by about 25%. iFIT can help facilitate fitting patients soon after amputation with the ability to be fit anywhere ...
-
Anteris reports successful interim results for the first-in-human trial for DurAVR
Anteris Technologies Ltd (ASX: AVR) reports the first-in-human DurAVR™ THV study met or exceeded its interim study objectives. At the 30-day follow up point, the first five patients of the planned 10-patient study showed: All five subjects continued to do well with no reported adverse events (no death, stroke, myocardial infarction, reintervention). An average 86% improvement in mean ...
-
Effects of prosthetic limb prescription on 3-year mortality among Veterans with lower-limb amputation
Method: Retrospective observational study on 4,578 Veterans hospitalized for LLA and discharged 2003- 2004, looking at time to all-cause mortality from the amputation surgical date up to the 3 yr anniversary. Results: 1,300 (28.4%) persons received a prescription for a prosthetic limb within 1 yr after the surgical amputation. About 46% (n = 2,086) of all Veterans w LLA died within 3 yr of the ...
-
CARMAT announces the completion of its investigations on previously identified quality issues
CARMAT (FR0010907956, ALCAR), the designer and developer of the world’s most advanced total artificial heart, aiming to fulfill an unmet medical need by providing a therapeutic alternative to people suffering from end-stage biventricular heart failure, announces the completion of investigations on its prostheses. Following the occurrence of a quality issue affecting some of its prostheses, ...
By Carmat
-
CARMAT voluntarily suspends Aeson implantations on a temporary basis
CARMAT (FR0010907956, ALCAR), the designer and developer of the world’s most advanced total artificial heart, aiming to fulfill an unmet medical need by providing a therapeutic alternative to people suffering from end-stage biventricular heart failure,announces that it has voluntarilysuspended, on a temporary basis, theimplants of its Aeson® artificial heart. Following the occurrence ...
By Carmat
-
Puzzle Medical announces partnership with Patent Attorney Scott Talbot from U.S. firm Cooley LLP
Puzzle Medical Devices Inc. is enthusiastic to partner with Patent Attorney C. Scott Talbot from Cooley LLP as it continues to grow its patent portfolio. Cooley LLP is the largest medical technology patent team in the US. Scott has more than two decades of experience in the medical technology industry, including with prosthetic heart valves, heart valve repair, orthopedic surgery, stents, blood ...
-
The Spark Center to house Epica International’s new HQ, warehousing, assembly facility
Epica International, a leader in cutting-edge mobile medical imaging systems and precision manufacturing robotics, unveiled their new headquarters and operations facility in Duncan on Thursday, May 13. The 27,000-square-foot headquarters, warehousing and assembly facility is located in The Spark Center on Spartanburg Community College’s Tyger River campus. Dr. Randy Axelrod, Epica CEO, ...
-
Epica International Announces Operations in Spartanburg, S.C. USA
Epica International, the leader in advanced ultra-high-resolution mobile medical imaging and robotic applications for human health, animal health and industrial enterprises, announces new corporate headquarters and operations in Spartanburg County, S.C. What: Epica International, with offices in San Clemente, California and manufacturing and R&D facilities in Florence and Pisa, Italy ...
-
Epica International Announces Operations in Spartanburg, S.C. USA
SPARTANBURG, SC – MAY 13, 2021 – Epica International, the leader in advanced ultra-high-resolution mobile medical imaging and robotic applications for human health, animal health and industrial enterprises, announces new corporate headquarters and operations in Spartanburg County, S.C. WHAT: Epica International, with offices in San Clemente, California and manufacturing and R&D ...
-
CARMAT receives FDA approval to use the new version of its artificial heart in the US Early Feasibility Study (EFS)
CARMAT (FR0010907956, ALCAR), the designer and developer of the world’s most advanced total artificial heart, aiming to fulfill an unmet medical need by providing a therapeutic alternative to people suffering from end-stage biventricular heart failure, today provides an update on its early feasibility study (EFS) in the United States. The FDA has granted CARMAT approval to use the new ...
By Carmat
-
Carmat outlines commercial and development plan for its total artificial heart
Addressable market in the bridge-to-transplant indication of more than 2,000 patients a year in Europe Company prepares for commercial launch in Q2 2021, with initial focus on Germany and France Robust clinical plan to support adoption and sales development Virtual conference with Stéphane Piat today at 5 pm CET CARMAT (FR0010907956, ALCAR), the designer and developer of the ...
By Carmat
-
APrevent signed the distribution agreement in Lithuania
APrevent announced on 7TH December that APrevent and Labostera signed the distribution agreement for APrevent® VOIS in Lithuania. We are going to provide patients with glottic insufficiency/unilateral vocal fold paralysis an innovative, safe and effective surgical treatment. Labostera was founded in year 2005, they are experienced in ENT surgical products including surgical voice ...
-
BioAesthetics Announces University Clinical Study for its Nipple Reconstruction Graft
BioAesthetics Corporation today announced a clinical study of its nipple reconstruction graft will be performed at Stanford Medicine beginning in 2021. The BioAesthetics® NACgraft™ biologic matrix is a decellularized skin allograft of the human nipple-areolar complex (NAC) intended to replace the NAC when lost due to mastectomy or other procedure. BioAesthetics hopes the NACgraft will ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you