Blood Bank News
-
Showcase
ALung Announces Commercial Development of its Breakthrough Next Generation Artificial Lung
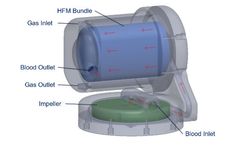
PITTSBURGH–(BUSINESS WIRE)– April 4, 2020 – ALung Technologies, Inc., the leading provider of low-flow extracorporeal carbon dioxide removal (ECCO2R) technologies for treating patients with acute respiratory failure, announced the recent initiation of commercial development of its next generation artificial lung, which expands the Company’s focus on highly efficient gas ...
-
Creative Diagnostics Launches New Anti-Small Molecule Label Antibodies to Facilitate NALFIA Development
Creative Diagnostics, a leading manufacturer and supplier of antibodies, antigens and assay kits, has recently announced a series of new Anti-Small Molecule Label Antibodies to facilitate the development of nucleic acid lateral flow immunoassay (NALFIA). These antibodies can specifically bind to small molecule labels and offer customers multiple choices, including Anti Digoxin monoclonal ...
-
Improve Staff Productivity
ZipThaw is a blood product thawer that uses revolutionary dry technology to quickly thaw blood products for patients in need. ZipThaw is dry, so there’s no messes like with water baths, and fast, so you can increase your thawed plasma throughput — it’s also easy for staffers to use. With simple thawing, staffers can spend their time elsewhere on other critical tasks. Last ...
-
Thaw More Plasma, Faster
Thaw More Plasma Faster with ZipThaw® Traditionally, plasma thawing is done in hot water baths which are slow, messy, and outdated. With their slow thawing to imprecise temperatures, time taken for warm up, and extensive maintenance and clean up needs, water baths make it impossible to maintain high plasma throughput levels. Now there’s an easier way to thaw more plasma, faster. ...
-
Time for a better way to thaw
For decades, medical professionals have used slow, cumbersome, imprecise water baths to thaw plasma. It’s time for a better way! Introducing ZipThaw: a dry, portable, rapid thawing device combined with our disposable ZipSleeve® anti-contamination barrier with heat sensors. As an alternative to water baths, dry plasma thawing is a groundbreaking innovation that unlocks numerous ...
-
CardioWise™ Receives ISO 13485:2016 Clearance for Design, Development, and Marketing of Software as a Medical Device (SaMD) For the Medical Device Industry
CardioWise, Inc., is pleased to announce that Perry Johnson Registrars, Incorporated has audited the CardioWise Quality Management System (QMS) and determined CardioWise is in conformance with ISO 13485:2016. Perry Johnson Registrars Certificate C2022-02910 was issued July 16, 2022, and represents the first step in the process of obtaining a CE mark and Medical Device Registration for the ...
-
CVRx Announces First Clinical Procedure with a New Ultrasound-Guided Implant Approach, the Latest Advancement in Barostim™ for the Treatment of Heart Failure Symptoms
MINNEAPOLIS, June 10, 2021 - CVRx©. developer of the world's first FDA-approved neuromodulation device to treat the symptoms of heart failure (HF). announced the completion of the first clinical procedure with the company's new lead implantation approach. The novel ultrasound-guided technique is the latest advancement of CVRx's Barostim™ Baroreflex Activation Therapy (BAT™) to ...
By CVRx
-
CardioWise Joins the Lucem Health Innovation Collaborative as a Founding Member
CardioWise® and Lucem Health™ announced that CardioWise has become a founding member of the Lucem Health Innovation Collaborative, a partner program designed to help AI/ML innovation move to the forefront of healthcare. The collaborative launched by Mayo Clinic along with other investing partners will help digital health innovators create, deploy, and commercialize transformational ...
-
CVRx Receives MR-Conditional Labeling Approval for its Barostim Heart Failure System
MINNEAPOLIS, MAY 9,2022 - CVRx, Inc. (NASDAQ: CVRX) ("CVRx"), developer of the world's first FDA-approved neuromodulation device to treat the symptoms of heart failure, has received U.S. Food and Drug Administration (FDA) approval for magnetic resonance (MR) conditional labeling for its Barostim System. The Barostim System now includes instructions to allow for safe MRI scans of the head and ...
By CVRx
-
CytoSorbents to Present at the Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum and Jefferies London Healthcare Conference
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces its participation in two upcoming investor conferences. CytoSorbents will present at the Canaccord Genuity Virtual MedTech, Diagnostics and Digital Health & Services Forum on Thursday, November 18, 2021. Dr. ...
-
CytoSorbents To Expand Direct Sales of CytoSorb to the United Kingdom, the Sixth Largest Medical Device Market in the World
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces that it has registered a new subsidiary, CytoSorbents Medical UK Limited, with plans to establish a direct sales presence within the United Kingdom (UK) and Ireland. CytoSorbents previously sold CytoSorb® via ...
-
CytoSorbents Receives Approximately $740,000 from the New Jersey Business Tax Certificate Transfer Program
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, today announced that it has received approximately $740,000, net of transactions costs, in cash proceeds from the sale of its 2020 Net Operating Loss (NOL) and R&D tax credits from the Technology Business Tax Certificate ...
-
Supira Medical, A Shifamed Portfolio Company, Closes $30m in an Oversubscribed Series C Financing
Company to Advance its Next Generation Percutaneous Ventricular Assist Device Los Gatos, Calif., February 28, 2022 – Supira Medical, Inc., a Shifamed portfolio company that is focused on developing a next-generation solution for temporary mechanical circulatory support, announced today the closing of $30M in Series C financing. Led by Cormorant Asset Management and The Capital Partnership ...
-
ExThera Medical & Fresenius Medical Care Sign Distribution Agreement for Seraph 100 Blood Purification Device in Mexico
Seraph 100 used for Blood Purification and Pathogen Reduction in Sepsis and COVID-19 Treatments MARTINEZ, Calif. – ExThera Medical and Fresenius Medical Care, the world’s leading provider of products and services for individuals with renal diseases, announced the expanded distribution of ExThera’s Seraph® 100 Microbind® Affinity Blood Filter in Mexico. In 2021, ExThera ...
-
T,C&A Lab Declares Capability of Biomedical Materials Testing
Since the last few decades, biomedical materials have been extensively used in a lot of medical applications, such as tissue scaffolds, wound care, regenerative dentistry, bone graft, implantable devices and cosmetic surgery. Earlier this month, T,C&A Lab, a branch of Alfa Chemistry, announces to provide biomedical material testing service for customers worldwide who would like to ensure ...
-
BioVentrix Expands Heart Failure Treatment Portfolio with Acquisition of MateraCor, Inc.
BioVentrix, Inc., a privately held medical device company focused on less invasive therapies to treat the left ventricle (LV), known to be the root cause of congestive heart failure (CHF), today announced that it has acquired MateraCor, Inc., a company focused on preventing progression of, and reversing, heart failure through the use of injectable alginate-based hydrogel. “This is an ...
-
Patient Enrollment is Complete in the DOD-Funded Study of Extracorporeal Hemopurification of Critically Ill COVID-19 Patients
MARTINEZ, Calif. – PURIFY OBS, an observational study of critically-ill COVID patients treated with the Seraph® 100 Microbind® Affinity Blood Filter (Seraph 100), has recently completed enrollment. A multicenter effort led by the US Department of Defense (DOD) published interim results in April 2021. Observations included dramatic improvement in survival and reduction in median ICU ...
-
Treatment for critically ill COVID patients results in significant improvement in survival rate according to new study
Patient survival doubles when Seraph 100 is used earlier during ICU stay MARTINEZ, Calif. – Critically ill patients treated with ExThera Medical’s Seraph® 100 Microbind® Affinity Blood Filter (Seraph 100) within 60 hours of ICU admission experienced a survival rate double that of patients treated after being in the ICU for 60 or more hours, according to a newly released ...
-
ExThera Medical and Asahi Kasei Medical Enter into Partnership to Expand Reach of Treatment for the Critically Ill
MARTINEZ, Calif. – ExThera Medical and Asahi Kasei Medical, a core operating company of the Asahi Kasei Group, have entered into an exclusive distribution agreement in Japan for ExThera’s Seraph® 100 Microbind® Affinity Blood Filter (Seraph 100). Per the agreement structure, ExThera Medical and Asahi Kasei Medical will collaborate to obtain regulatory approval of Seraph 100 ...
-
CytoSorbents Appoints Daniel Wendt, MD, PhD, MHBA, FECTS as Vice President, Medical Affairs Cardiovascular
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces the appointment of Professor Daniel Wendt, MD, PhD, MHBA, FETCS as Vice President, Medical Affairs Cardiovascular commencing January 1, 2022. Dr. Wendt is an internationally renowned academic cardiac surgeon with over ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you