Medtech News
-
Showcase
PhotoniCare Announces FDA Clearance for First-In-Class Technology for Imaging the Ear
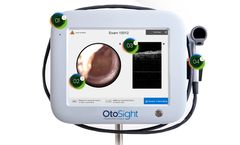
Champaign, Ill. – Jan. 6, 2020 – PhotoniCare, Inc., a company dedicated to revolutionizing healthcare by providing healthcare providers with better diagnostic tools, today announced that the U.S. Food & Drug Administration (FDA) has granted 510(k) clearance for its TOMi™ Scope [OtoSight Middle Ear Scope] for non-invasive imaging of the middle ear. Using optical coherence ...
-
Caresyntax Announces Launch of Novel Real World Evidence (RWE) Program and Software Portfolio of Digital Catalysts for Commercial Acceleration of Medtech Partners
Caresyntax, the leading vendor-neutral surgical data and intelligence platform converging AI-powered software, devices, and clinical services, today announced the launch of Caresyntax Clinical Data as a Service (CDaaS) for Medtech industry partners. The launch of Caresyntax CDaaS follows the recently announced addition of CQInsights (CQI) and appointment of Dr. Bruce Ramshaw as Chief Medical ...
By Caresyntax
-
Vitalograph nominated as finalist in the 2023 Irish Medtech Awards
The awards are run by the Irish Medtech Association and are part of the Medtech Rising Business Leaders Conference, which will be held in Galway on December 7th. The vision of the Irish Medtech Association is to strongly position Ireland as a global leader in innovative patient-centred medical technology solutions, helping to set the future global healthcare agenda, with a proven ecosystem that ...
-
Stefan Blomsterberg appointed as new CEO of Cellink Bioprinting AB
Stefan Blomsterberg has been appointed as the new CEO of CELLINK. Cecilia Edebo, current CEO, requested earlier this year to resign from her position to pursue a new career opportunity outside the BICO Group. At the time of her resignation, a recruitment process was initiated which concluded in the appointment of Stefan Blomsterberg. Stefan Blomsterberg (born 1964) brings 20+ years of experience ...
By CELLINK
-
CELLVIE wins the prestigious MedTech Innovator Value Award
MedTech Innovator is the leading program for medical devices, digital health and diagnostic companies and is the largest accelerator for life science companies globally. Every year, start-ups from all over the world are invited to present their solution and vision for a chance to be selected to join the MedTech Innovator Accelerator. The program culminates in a competition for a grand prize of ...
By cellvie Inc.
-
SpinTech CEO Presents STAGE at MedTech Strategist Innovation San Francisco 2021
Ward Detwiler, our President & CEO, had the opportunity to speak about how STAGE is innovating the world of MR at MedTech Strategist Innovation 2021. ...
-
electroCore to Participate at Canaccord Genuity MedTech and Diagnostics and Digital Health & Services Forum
electroCore, Inc. (Nasdaq: ECOR), a commercial-stage bioelectronic medicine company, today announced that electroCore’s Chief Executive Officer, Dan Goldberger, will be conducting one-on-ones at the Canaccord Genuity MedTech and Diagnostics and Digital Health & Services Forum. Canaccord Genuity MedTech and Diagnostics and Digital Health & Services Forum Date: Thursday, November ...
-
Pulmonx to Participate at Upcoming Investor Conferences
Pulmonx Corporation (Nasdaq: LUNG) (“Pulmonx”), a global leader in minimally invasive treatments for severe lung disease, today announced the company will be participating in two upcoming investor conferences in New York. Pulmonx management is scheduled to participate in a fireside chat at the Stifel Healthcare Conference on Wednesday, November 16, 2022, at 8:00AM ET. Interested ...
-
Elucid Appoints Dr. Michael Lesh to Board of Directors
BOSTON, November 3, 2022 – Elucid, Inc., a leading medical technology company providing physicians with AI-powered imaging analysis software to characterize cardiovascular disease, today announced the addition of Michael D. Lesh, MD, FACC, to its Board of Directors. “We look forward to what Dr. Lesh will bring to the board with his extensive background as a cardiologist, medtech ...
By Elucid
-
Quantum-Si to Participate in the 16th Annual Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum
Quantum-Si Incorporated (Nasdaq: QSI) (“Quantum-Si,” “QSI” or the “Company”), a life sciences company commercializing the first-next generation single-molecule protein sequencing platform, today announced that it will be participating in the 16th Annual Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum taking place at the Westin New ...
-
Inari Medical to Participate in the Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum
Inari Medical, Inc. (NASDAQ: NARI) (“Inari”), a medical device company focused on developing products to treat and transform the lives of patients suffering from venous and other diseases, announced today that its management team will participate in the Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum at the Westin NY Grand Central Hotel in New York on ...
-
Anuncia Medical, Inc., receives FDA clearance for the ReFlow System Mini for the treatment of patients with hydrocephalus
Anuncia Medical, Inc., announced that the U.S. Food and Drug Administration (FDA) cleared the ReFlow® System Mini for the treatment of patients with hydrocephalus and other cerebrospinal fluid (CSF) disorders that require shunting. Anuncia Medical announced FDA clearance of ReFlow® System Mini for the treatment of cerebrospinal fluid (CSF) disorders. The FDA 510(k) clearance allows ...
-
Tissium Appoints Sara Toyloy as Independent Board Member
TISSIUM, a privately-owned medtech company developing biomorphic programmable polymers for tissue reconstruction, announced today it has appointed Sara Toyloy as its fourth independent member of the Board of Directors. Ms. Toyloy has over 30 years of experience in the medical device industry. In 2020, she founded Fabrica Consulting LLC, a medical device consulting firm providing global ...
By TISSIUM
-
Transformative Dementia and Alzheimer’s treatment company Kurve Therapeutics launches $50m equity crowdfunding raise
Next-generation Medtech innovator Kurve Therapeutics begins an equity crowdfunding journey with its $50 million Regulation A+ raise. Private investors can now invest in a company aiming to disrupt and transform a $1.3 trillion dementia market alone while benefiting millions living with neurodegenerative diseases. Diseases like Dementia and Alzheimer’s afflict millions of people across the ...
-
iSTAR Medical appoints leading glaucoma specialist Prof. Kaweh Mansouri as Chief Medical Officer
WAVRE, Belgium — 6 September 2022: iSTAR Medical, a medtech company delivering breakthrough eye care solutions to patients, today announces the appointment of Dr Kaweh Mansouri as Chief Medical Officer (CMO). Dr Mansouri is a medical and surgical eye specialist, sub-specializing in glaucoma and cataract. Currently a Consultant Ophthalmologist at the Glaucoma Center, Swiss Visio, ...
-
The HealthTech For Care Innovation Award will be presented for the first time at the HealthTech Innovation Days on October 13 and 14, 2022 in Paris.
25 European companies from the Biotech, Medtech, Innovative Services & Digital Health sectors, committed to patients, selected by the European clusters, will present their innovation to a jury composed of HTFC’s sponsors and the HTID health innovation ecosystem. The HealthTech Innovation Days (HTID®) event, organized by the HealthTech For Care (HTFC) endowment fund, is launching ...
-
Confluent Medical Technologies Announces Grand Opening of Costa Rica Expansion
Confluent Medical Technologies (Confluent), today announced the Grand Opening of the latest addition to their large scale manufacturing center of excellence in Alajuela, Costa Rica expanding their capacity for Nitinol processing and catheter manufacturing. This new facility will be co-located with the existing Confluent Costa Rica facility and will add an additional 66,000 square feet to this ...
-
Kurve Therapeutics, Innovative Medtech and Specialty-Drug Company Focused on Treating Neurodegenerative Diseases, Files Regulation A Offering
Kurve Therapeutics, a next-generation medtech innovator focused on developing treatments for neurodegenerative diseases, announced it has filed a Form 1-A preliminary offering circular with the U.S. Securities and Exchange Commission (SEC) for a $50 million funding round [kurvetx.com/offering-circular]. Investment opportunities are now open to public investors for offers starting at $580 for 100 ...
-
BioSerenity, Inc. appoints new leadership in North America
BioSerenity, a high-growth company specializing in research and remote diagnostic solutions for neurology, sleep medicine, and cardiology, announces Jeff Wren’s appointment as Chief Executive Officer of North America. Jeff Wren assumes the leadership role in the states as Pierre Frouin, CEO of BioSerenity, moves back to Europe. Since its expansion to the United States in 2019, ...
By BioSerenity
-
Linear Health Sciences’ Early Success Highlighted at SEMDA Medical Device Conference
ATLANTA, Georgia – The early success of Linear Health Sciences in developing its Orchid™ Safety Release Valve to prevent unwanted dislodgement of IV catheters was highlighted in a panel discussion at the recent annual conference of the Southeastern Medical Device Association (SEMDA). Dan Clark, co-founder and Chief Marketing Officer of Linear Health Sciences, represented the company ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you