Cardiopulmonary News
-
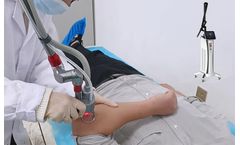
The CO2 fractional laser skin resurfacing in action
The best CO2 fractional laser machine is a laser treatment for spots, wrinkles, scars and warts, which is usually a type of laser that stimulates the skin tissue through the focused energy emitted by the gas laser. Which not only blasts the melanin particles on the surface instantly, but also stimulates the collagen tissue underneath the skin to gradually repair the skin. It also stimulates the ...
-
Newly Published Study Proves Value and CliNewly Published Study Proves Value and Clinical Benefit of Daxor’s Blood Volume (BVA-100) Diagnostic in the Evaluationnical Benefit of Daxor’s Blood Volume (BVA-100) Diagnostic in the Evaluation of Heart Failure
Daxor Corporation (Nasdaq: DXR), the global leader in blood volume measurement technology, today announces a new study in the Journal of Cardiovascular Translational Research that examined pressure-volume profiles in heart failure patients using Daxor’s BVA-100 blood test to measure intravascular volume overload compared to a commercially available implantable pulmonary artery diastolic ...
-
Aerami Therapeutics to Present at the LifeSci Partners Summer Symposium
Aerami Therapeutics, Inc. (the “Company”) announced today that Anne Whitaker, Chief Executive Officer, will present a corporate overview at the Private Healthcare Company Virtual Summer Symposium, hosted by LifeSci Partners, on August 4 and 5, 2020. Please click the following link to register Presentation Details: Date: August 5, 2020 Time: 9:00 am Eastern Time About Aerami ...
-
Aerami Therapeutics Announces Completion of Dosing in Phase 1 Trial of AER-901 (inhaled imatinib), a Critical Milestone Toward the Planned Initiation of a Phase 2 Trial in Pulmonary Arterial Hypertension (PAH) During the First Half of 2023
AER-901 (inhaled imatinib), a potential reverse-remodeling therapy, is being developed to address unmet need for PAH treatments that target pulmonary vascular remodeling while supporting ease-of-use for patients. PAH is a rare and progressive disease with no cure; pulmonary vascular remodeling is believed to drive disease progression and poor outcomes, however, all currently approved therapies ...
-
Aerami Therapeutics Appoints Lisa Yanez as Chief Executive Officer
Aerami Therapeutics Holdings, Inc (“Aerami”), a clinical-stage biopharmaceutical company dedicated to breathing life into the treatment of serious cardiopulmonary and cardiometabolic conditions, today announced the appointment of Lisa Yañez as chief executive officer, effective January 1, 2023. As part of a planned leadership transition, Steve Thornton will retire as CEO at the ...
-
Third Pole Therapeutics Receives $32M Strategic Equity Investment to Accelerate its Tankless Inhaled Nitric Oxide (iNO) Platform
Third Pole Therapeutics, a privately held company developing critical life-sustaining cardio-pulmonary therapies, announced today a $32M equity investment, from a large medical device innovator. Bill Athenson, CEO of Third Pole, stated, “We are thrilled to receive this investment, which raises Third Pole Therapeutics’ series B financing and commitments to date to over $85M. I am ...
-
Assessing Heart Function and Fluid Status with Cardiac Scales: Interview with John Lipman, CEO, and Corey Centen, Founder, Bodyport
Bodyport, a medtech company based in San Francisco, developed the Bodyport Cardiac Scale, a set of weighing scales that can non-invasively assess fluid status and heart function. The scales can detect these signals through the feet when a patient steps onto the device. The technology is intended to be convenient and requires only 20 seconds each morning, and should easily lend itself to ...
-
Adagio Medical Announces Publication of Cryocure-2 Data Reporting 85% Freedom from AF at 12 Months After a Single Ablation Procedure in Patients with Persistent Atrial Fibrillation and Discusses Current Status of iCLAS Cryoablation System
Adagio Medical, Inc., a leading innovator in catheter ablation technologies for atrial fibrillation (AF) and ventricular tachycardia (VT), announced the publication of the results of its Cryocure-2 ultra-low temperature cryoablation (ULTC) study in patients with AF in the Journal of American College of Cardiology (JACC), Clinical Electrophysiology.1 The study prospectively enrolled 79 patients at ...
-
Endotronix Announces Positive Data from SIRONA 2 in Late-Breaking Clinical Trial Session
Endotronix, Inc., a digital health and medical technology company dedicated to advancing the treatment of heart failure (HF), today announced positive data from the SIRONA 2 clinical trial evaluating safety and efficacy of its Cordella™ Pulmonary Artery Pressure Sensor System (Cordella Sensor) in New York Heart Association (NYHA) class III heart failure patients. The prospective, ...
-
First Patients in the U.S. Enrolled in Biosense Webster Clinical Study To Evaluate VARIPULSE™ Catheter for Atrial Fibrillation
Johnson & Johnson MedTech* announced today that Biosense Webster, Inc., a worldwide leader in the science and technology of cardiac arrhythmia treatment, enrolled the first patients in its admIRE clinical study in the United States. admIRE is a prospective, multi-center, non-randomized study that will enroll more than 400 patients in the U.S. to evaluate the safety and effectiveness of ...
-
CytoSorbents to Present at the Canaccord Genuity MedTech, Diagnostics and Digital Health & Services Forum and Jefferies London Healthcare Conference
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces its participation in two upcoming investor conferences. CytoSorbents will present at the Canaccord Genuity Virtual MedTech, Diagnostics and Digital Health & Services Forum on Thursday, November 18, 2021. Dr. ...
-
CytoSorbents To Expand Direct Sales of CytoSorb to the United Kingdom, the Sixth Largest Medical Device Market in the World
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, announces that it has registered a new subsidiary, CytoSorbents Medical UK Limited, with plans to establish a direct sales presence within the United Kingdom (UK) and Ireland. CytoSorbents previously sold CytoSorb® via ...
-
Stanford Physicians Continue to Rely on EchoPixel’s Technology
EchoPixel has had a long-standing relationship with Stanford physicians, who were early adopters of its technology, beginning with the successful surgery of the conjoined Sandoval twins in 2017. The affiliation has continued in partnership with Michael Ma, MD, Assistant Professor of Cardiothoracic Surgery in the Division of Pediatric Cardiac Surgery at Stanford University, who has used ...
-
Kardium’s Globe® System Begins Clinical Trial at St. Paul’s Hospital
VANCOUVER, British Columbia –?Kardium?Inc.?announces a successful first-in-BC investigational* clinical procedure using the Globe® Mapping and Ablation System. Dr. Marc Deyell of St. Paul’s Hospital leads the study using the Globe System to navigate inside the heart and diagnose where the rhythm disturbances occur. Once identified, the same device can then deliver immediate ...
By Kardium Inc.
-
CytoSorbents Receives Approximately $740,000 from the New Jersey Business Tax Certificate Transfer Program
CytoSorbents Corporation (NASDAQ: CTSO), a leader in the treatment of life-threatening conditions in the intensive care unit and cardiac surgery using blood purification, today announced that it has received approximately $740,000, net of transactions costs, in cash proceeds from the sale of its 2020 Net Operating Loss (NOL) and R&D tax credits from the Technology Business Tax Certificate ...
-
Xeltis accelerating clinical trial program
Xeltis announced today that is planning to accelerate its clinical trial program for three restorative devices in parallel. CEO Eliane Schutte provided a sneak preview on Xeltis’ plans to an audience of investors and medtech companies at the Medtech and Diagnostics Forum organised by the investment bank Canaccord Genuity Capital Markets. Xeltis currently has three restorative devices in ...
By XELTIS BV
-
Study Demonstrates Importance and Clinical Utility of Daxor’s Blood Volume (BVA-100) Diagnostic in the Assessment of Heart Failure
Daxor Corporation (Nasdaq: DXR), the global leader in blood volume measurement technology, today announces new data demonstrating the importance and clinical utility of Daxor’s BVA-100 blood test in heart failure (HF) patients published in the Journal of Cardiac Failure. The results of the study show that Daxor’s BVA-100 provides unique and actionable data that is critical for ...
-
Third Pole Therapeutics Closes $25M Financing - eNOfit Wearable Device Now Ready for Clinical Trials
eNOfit™, Third Pole’s novel nitric oxide mobile-wearable device designed to provide “on-the-go” treatment for patients suffering from severe COPD and ILD, to enter clinical trials in the second half of this year Funding will support device optimization and clinical trials; Pivotal trial data expected in the second half of 2023 Third Pole Therapeutics, a privately ...
-
Third Pole Therapeutics to Participate in H.C. Wainwright BioConnect and BIO Partnering at JPM during J.P. Morgan 40th Annual Healthcare Conference
Third Pole Therapeutics, a privately held company developing critical life-sustaining cardio-pulmonary therapies, announced today that it is presenting at the H.C. Wainwright BioConnect 2022 Conference and participating in BIO Partnering at JPM. All events are virtual and scheduled concurrently with the J.P. Morgan 40th Annual Healthcare Conference. Details of the events and presentation are as ...
-
Endotronix Announces FDA Approval for PROACTIVE-HF Pivotal Trial Design Change to Single-Arm Study
Endotronix, Inc., a digital health and medical technology company dedicated to advancing the treatment of heart failure (HF), today announced the U.S. Food and Drug Administration (FDA) has granted approval for an amendment to the company’s PROACTIVE-HF study, a pivotal Investigational Device Exemption (IDE) trial investigating the Cordella Pulmonary Artery (PA) Pressure Sensor1, shifting ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you