ENT News
-
Showcase
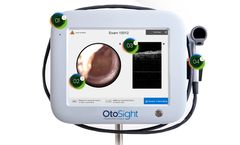
PhotoniCare Announces FDA Clearance for First-In-Class Technology for Imaging the Ear
Champaign, Ill. – Jan. 6, 2020 – PhotoniCare, Inc., a company dedicated to revolutionizing healthcare by providing healthcare providers with better diagnostic tools, today announced that the U.S. Food & Drug Administration (FDA) has granted 510(k) clearance for its TOMi™ Scope [OtoSight Middle Ear Scope] for non-invasive imaging of the middle ear. Using optical coherence ...
-
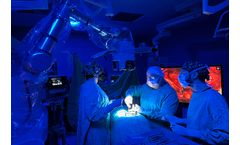
Synaptive Medical Receives FDA 510(k) Clearance for Near-Infrared Fluorescence Visualization, Expanding Application of Existing Robotic Exoscope
Near-Infrared fluorescence now available on Modus X robotic exoscope, complementing system’s existing advanced fluorescence capabilities New Infrared fluorescence enables the use of Modus X for visualization of vessels in conjunction with indocyanine green New patented 4K ICG Image Fusion for real-time contrast of microsurgical vessels against background anatomy Synaptive Medical ...
-
Synaptive Medical Welcomes Chris Marrus as New Chief Commercial Officer
Synaptive Medical Inc., a global med tech and technology company solving surgical, imaging and data challenges, is proud to announce the appointment of Chris Marrus as Chief Commercial Officer. This appointment underscores Synaptive’s commitment to driving sales, expanding its market presence, and advancing innovative solutions in imaging and surgical technology focused on combining the ...
-
Impel Pharmaceuticals Announces Filing of Voluntary Chapter 11 Cases and Signing of `Stalking Horse` Agreement to Facilitate Sale
Patient Access to Trudhesa® to Continue Uninterrupted Sufficient Liquidity to Fund Day to Day Operations During Court-Supervised Process Impel Pharmaceuticals Inc. (OTCQX: IMPL) ("Impel" or "the Company"), a commercial-stage biopharmaceutical company with a mission to develop transformative therapies for people suffering from diseases with high unmet medical needs, today announced that ...
-
Synaptive Medical Introduces Cutting-Edge Modus X Robotic Exoscope in Europe with its First Imstallation at Azienda Ospedaliera Universitaria Federico ll in Naples, Italy
Synaptive Medical Inc., a global medical technology innovator, is pleased to announce the inaugural installation of its next-generation robotic exoscope, Modus X in Europe. This highly anticipated exoscope delivers a leap forward for surgical visualization in neurosurgery, ENT, plastics, and spine procedures, and it has made its European debut at Azienda Ospedaliera Universitaria Federico II in ...
-
Veersing Effect - How Intra-Oral Scanners Introduce Errors in Edentulous Jaws and Flat Planes
Dentate cases can introduce errors in your models due to refraction, reflection, and light transmission right through enamel. The asymmetric tooth morphologies can help your scanner stay on track. Flat/edentulous plane/ridges introduce a whole other set up challenges. The main one being the Veersing Effect where scanners incorrectly plot data where it doesn’t belong. This is reproduceable ...
By CAD-Ray
-
Tris Pharma Presents Data Reinforcing Limited Abuse Potential of Investigational Pain Therapy Cebranopadol at the 2023 PAINWeek Conference
Positive results confirm findings from prior studies evaluating the abuse potential of cebranopadol, further highlighting its potential as a safe and potent alternative to traditional opioids Registrational phase 3 clinical trials of cebranopadol in moderate to severe acute pain are on track to initiate in Q4 2023 – Business Wire – Tris Pharma, Inc. (Tris), a commercial-stage ...
-
Bayer receives U.S. FDA Fast Track Designation for asundexian atrial fibrillation program
The OCEANIC-AF (atrial fibrillation) study with asundexian is part of the Phase III OCEANIC clinical trial program enrolling more than 27,000 patients in over 40 countries Asundexian is being evaluated as a potential improved treatment option in stroke prevention and could be part of an entirely new class of treatment options in thrombosis management that aims to uncouple efficacy from ...
By Bayer AG
-
Peer reviewed publications reporting the effectiveness of the Kurve intranasal insulin product
Nasaldrugdeliveryblog.com opens lines of communication between drug delivery researchers, device manufacturers. The first blog to address issues related to nasal drug delivery formulation, research, delivery, and devices was launched by Kurve Technology, Inc., a leading developer of nasal drug delivery devices. The blog - http://www.nasaldrugdeliveryblog.com - invites scientists, researchers ...
-
Infrared Coagulation of The Inferior Turbinate: A new treatment for refractory chronic rhinitis
Nasal obstruction remains a challenge to the modem day rhinologist. In the evolving climate of "cost containment" and "risk-outcome analyses", office-based inferior turbinate reduction has regained popularity. This article describes a new method to reduce nasal obstruction in patients with chronic rhinitis when conventional medical therapy has failed. Rhinitis is chronic (CR) when symptoms of ...
-
New Surgical Images
As an organisation, we receive surgical images from a variety of specialities from surgeons all over the world. We are very grateful for this: it helps us to continuously develop medical devices designed for the need surgeons have, as well as confirming that the designs we have launched perform as intended. The images form a library of knowledge and education, and we have recently donated some ...
-
Combines mesenchymal stem-cell-based product, with Kurve’s intranasal drug delivery technology
Orgenesis Inc. (NASDAQ: ORGS) (“Orgenesis” or the “Company”), a global biotech company working to unlock the full potential of cell and gene therapies, and Kurve Therapeutics, a next-generation medtech and drug formulation company, today announced positive preclinical study results for intranasal administration of a cell-based oncolytic virus bearing product. The results ...
-
Candida auris and Infection Control Resources for Healthcare Providers
Candida auris is an emerging multidrug-resistant microorganism that was first identified in 2009 in Asia. It has quickly become a cause of severe infections around the world, including in the United States according to the Centers for Disease Control and Prevention (CDC). It is as yeast, which is a type of fungi. CDC describes Candida auris as a concerning drug-resistant fungus because it is: ...
-
BRIM Biotech submits US Phase 3 clinical trial protocol for BRM421 in the treatment of dry eye disease to the FDA
BRIM Biotechnology, Inc. (“BRIM,” TPEx 6885) announced today that it has officially submitted the Phase 3 clinical trial protocol for BRM421 in the treatment of dry eye disease (DED) to the U.S. Food and Drug Administration (FDA). If the FDA has no further comments after the 30-day review period, BRIM will proceed to initiate the Phase 3 clinical trial in December. Based on its ...
-
VeriSIM Life Named “Predictive Analytics Solution of the Year” by BioTech Breakthrough
VeriSIM Life (VeriSIM) www.verisimlife.com, the leading artificial intelligence (AI)-enabled, unique R&D decisions de-risker for breakthrough drug development, today announced it has been selected as the winner of the “Predictive Analytics Solution of the Year" in the BioTech Breakthrough Awards program conducted by BioTech Breakthrough. Biotech Breakthrough is a leading independent ...
By VeriSIM Life
-
Intrommune Therapeutics Completes Enrollment in the Phase 1 OMEGA Study for Peanut Allergy
Observed only mild and transient adverse events, with no use of emergency epinephrine Observed 98.4% patient compliance to date with novel OMIT therapeutic modality Safely started patients on dose 4, potentially reducing the time to reach maintenance by several weeks NEW YORK, Oct. 26, 2022 (GLOBE NEWSWIRE) -- Intrommune Therapeutics, Inc., a New York-based, clinical stage biotechnology ...
-
miDiagnostics to pilot innovative, fast COVID-19 PCR testing solutions at Brussels Airport
In the past year miDiagnostics has been working on the development of an ultra-fast PCR test based on imec technology, to detect COVID-19, with a test result in less than 15 minutes. In cooperation with Brussels Airport, this nasal-swab ultra-fast PCR test, as well as the new breath sampler developed by imec and licensed by miDiagnostics, will be tested in practice during a study at the ...
-
Dr. Davies-Cutting Brings Industry Leading Technical Expertise to Nob Hill Therapeutics
Albuquerque. NM, Nob Hill Therapeutics announced today the appointment of Craig Davies-Cutting, PhD, as Vice President of Research and Development. Dr. Davies-Cutting will contribute his extensive respiratory product development and industry expertise to the development and commercialization of Nob Hill Therapeutics’ first-of-its-kind DryNeb dry powder nebulizer inhalation drug delivery ...
-
Enalare Therapeutics Receives Orphan Drug Designation From the U.S. FDA on ENA-001 for the Treatment of Apnea of Prematurity (AoP)
ENA-001, a New Chemical Entity (NCE) with a novel mechanism of action as an agnostic respiratory stimulant, has previously been granted Rare Pediatric Disease designation for the Treatment of AoP by the FDA, with eligibility for a priority review voucher (PRV). The compound is also being developed by Enalare for the treatment of post-operative respiratory depression and community drug overdose. ...
-
Myrtelle and Forge Biologics Announce Viral Vector and Plasmid DNA cGMP Manufacturing Partnership
Myrtelle Inc., (Myrtelle), a clinical stage gene therapy company focused on developing transformative treatments for neurodegenerative diseases, and Forge Biologics, a gene therapy-focused contract development and manufacturing organization, today announced a manufacturing partnership that will advance Myrtelle’s novel gene therapy for monogenic hearing loss, Myr-201, into clinical trials ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you