Internal Medicine News
-
Bayer driving innovation in radiology with new research across the portfolio presented at ECR
Featured presentations include development compound gadoquatrane, a novel contrast agent for MRI Further research explores potential of AI and digital advances in medical imaging Bayer announced today that new data from the company’s advanced radiology portfolio will be presented at the 2023 European Congress of Radiology (ECR), taking place from March 1-5 in Vienna, Austria. The ...
By Bayer AG
-
xCures partners with RenovoRx to assist patients in accessing RenovoRx’s TIGeR-PaC study
xCures, Inc. is proud to announce their partnership with RenovoRx, a clinical-stage biopharmaceutical company that focuses on fighting cancer through localized treatment of difficult-to-treat tumors, such as pancreatic adenocarcinoma. Through this collaboration, xCures will help eligible pancreatic cancer patients access a Phase III clinical study entitled, Targeted Intra-arterial Gemcitabine vs. ...
By xCures
-
LISCure`s Phase 2 Drug Candidate Granted Orphan Drug Designation (ODD) from US FDA
LB-P8, a novel drug being developed by LISCure Biosciences, Inc. (LISCure) as a treatment for Primary Sclerosing Cholangitis (PSC), has been designated as an orphan drug by the US FDA. On December 1, 2022, the US FDA notified LISCure of the orphan drug designation of LB-P8, LISCure's microbiome novel drug, as treatment of PSC. LB-P8 is, to the best of company's knowledge, the first drug to be ...
-
LISCure Biosciences Announces Oral Presentation of New Drug Candidate for NASH at Upcoming AASLD The Liver Meeting 2022
LISCure Biosciences, a clinical-stage biopharmaceutical company developing microbe-oriented new drugs based on innovative technologies, today announced that a new drug candidate for NASH was selected for an oral presentation at the American Association for The Study of Liver Diseases (AASLD) The Liver Meeting 2022, to be held November 4-8 in Washington DC. As part of the presentation, LISCure ...
-
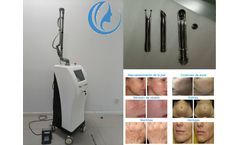
What is CO2 Fractional Laser Treatment
CO2 fractional laser resurfacing machine treatment can quickly improve skin texture, firm skin, improve large pores, and make skin smooth and tender like water. CO2 fractional laser technology is now the world's leading technology for wrinkle removal and skin rejuvenation. Through pulsed fractional laser, laser beauty has been raised to a new level. Bvlaser is a professional CO2 fractional laser ...
-
FDA Approves Taiho’s LYTGOBI® (futibatinib) Tablets for Previously Treated, Unresectable, Locally Advanced or Metastatic Intrahepatic Cholangiocarcinoma
LYTGOBI (pronounced “light-GOH-bee”) delivered an objective response rate of 42% and median duration of response of 9.7 months in the primary analysis of the pivotal clinical trial. LYTGOBI covalently binds to FGFR2 and inhibits the signaling pathway. The other approved FGFR inhibitors are reversible ATP-competitive inhibitors. LYTGOBI previously received breakthrough, orphan drug ...
-
Taiho Oncology Announces Updated Efficacy and Safety Data for Futibatinib in Cholangiocarcinoma at 2022 ASCO Annual Meeting
PRINCETON, N.J., June 3, 2022 – Taiho Oncology, Inc. announced today updated results of the Phase 2 FOENIX-CCA2 trial of futibatinib, confirming results observed in an earlier analysis. The trial was conducted in patients with locally advanced or metastatic unresectable intrahepatic (inside the liver) cholangiocarcinoma (iCCA) harboringFGFR2 gene rearrangements including fusions. These data ...
-
Affimed Reports First Quarter 2022 Financial Results and Highlights Recent Operational Progress
AFM13 monotherapy: Completed enrollment in the REDIRECT study for patients with relapsed or refractory PTCL; on track to report topline data in the fourth quarter of 2022 AFM13 combination with NK cells: Reported data at AACR 2022 showing a 100% objective response rate (ORR) and an increase in complete responses (CR) from 38%, reported in December, to 62% after a second cycle of therapy in 13 ...
By Affimed GmbH
-
IDK Stool Preparation Systems
IDK® would like to support you and help you improve processes: our stool preparation systems will be color-coded from now on in order to better distinguish between the different extraction buffers. This will help to eliminate any confusion or uncertainty. White label: IDK Extract® (K 6999) Stool preparation system filled with the extraction buffer IDK Extract® to be used to analyse ...
-
Cara Therapeutics to Present at Upcoming Investor Conferences
Cara Therapeutics, Inc. (Nasdaq: CARA), an early commercial-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced that Christopher Posner, President and Chief Executive Officer, will present at the following investor conferences: H.C. Wainwright Global Investment Conference Wednesday, May 25, 2022, at 10:30 a.m. ...
-
Kapruvia® approved by European Commission for the treatment of moderate-to-severe pruritus in hemodialysis patients
First approved therapy in Europe for the treatment of chronic kidney disease (CKD)-associated pruritus in hemodialysis patients First launches in Europe expected in H2 2022 Vifor Fresenius Medical Care Renal Pharma (VFMCRP) and Cara Therapeutics, Inc. (Nasdaq: CARA) today announced that the European Commission has granted marketing authorization to Kapruvia® (difelikefalin) for the ...
-
Creative Enzymes Probiotics Expands Service Offering with the Launch of Lactococcus Strains
Creative Enzymes, a professional and experienced supplier of enzymes, has a great reputation in the industry. With the continuous expansion of business areas and scale, the company has set foot in other areas and established a sound probiotic production and supply chain. These days, Creative Enzymes Probiotics is excited to announce the launch of Lactococcus strains. This will further enrich the ...
-
Cara Therapeutics to Present at the 21st Annual Needham Virtual Healthcare Conference
Cara Therapeutics, Inc. (Nasdaq: CARA), an early commercial-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced that Christopher Posner, President and Chief Executive Officer, will participate in a fireside chat at the 21st Annual Needham Virtual Healthcare Conference on Monday, April 11, 2022, at 1:30 p.m. ET. ...
-
Cara Therapeutics Announces Biomarker Data from KALM-1 and KALM-2 Trials of KORSUVA (difelikefalin) Injection Selected for Presentation at National Kidney Foundation Spring Clinical Meetings 2022
Cara Therapeutics, Inc. (Nasdaq: CARA), an early commercial-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced biomarker data from the KALM-1 and KALM-2 clinical trials evaluating KORSUVA™ (difelikefalin) injection for the treatment of chronic kidney disease-associated pruritus in patients undergoing ...
-
U.S. Food and Drug Administration (FDA) Accepts for Priority Review Taiho Oncology’s New Drug Application for Futibatinib for Cholangiocarcinoma
PRINCETON, N.J., MARCH 30, 2022 – Taiho Oncology, Inc. and Taiho Pharmaceutical Co., Ltd. announced today that the U.S. Food and Drug Administration (FDA) has accepted for priority review the New Drug Application (NDA) for futibatinib in the treatment of patients with previously treated locally advanced or metastatic cholangiocarcinoma (CCA) harboringFGFR2 gene rearrangements, including ...
-
Cara Therapeutics Announces Oral KORSUVA (difelikefalin) Improves Itch and Inflammatory Biomarkers in Atopic Dermatitis Patients with Moderate-To-Severe Pruritus
Cara Therapeutics, Inc. (Nasdaq: CARA), an early commercial-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced data from a sub-study of the KARE Phase 2 clinical trial demonstrated Oral KORSUVA™ (difelikefalin) improved itch and inflammatory biomarkers in atopic dermatitis (AD) patients with ...
-
Cara Therapeutics Announces Oral KORSUVA (difelikefalin) Data Selected for Late-Breaking Presentation at 2022 American Academy of Dermatology Annual Meeting
Oral presentation will include biomarker data from KARE Phase 2 clinical trial of Oral KORSUVA™ (difelikefalin) for the treatment of moderate-to-severe pruritus in atopic dermatitis patients Oral KORSUVA (difelikefalin) Phase 3 pruritus programs in non-dialysis dependent advanced chronic kidney disease and atopic dermatitis, respectively, to be discussed during Company’s virtual ...
-
Cara Therapeutics to Present at May Investor Conferences
Cara Therapeutics, Inc. (Nasdaq: CARA), an early commercial-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced that Christopher Posner, President and Chief Executive Officer, will present at the following investor conferences in May: BofA Securities 2022 Healthcare Conference Tuesday, May 10, 2022, at 2 p.m. ...
-
Kapruvia® receives positive CHMP opinion for the treatment of moderate-to-severe pruritus in hemodialysis patients
Committee for Medicinal Products for Human Use (CHMP) recommends approval of Kapruvia® (difelikefalin) as first therapy in Europe for the treatment of chronic kidney disease associated-pruritus (CKD-aP) in hemodialysis patients European Commission decision for EU Marketing Authorization is expected in Q2 2022 Kapruvia® is approved in the U.S. under the trade name KORSUVA™ ...
-
Cara Therapeutics to Host Virtual R&D Day on March 11, 2022
Cara Therapeutics, Inc. (Nasdaq: CARA), an early commercial-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced the Company will host a virtual research and development (R&D) event on Friday, March 11, 2022, from 1:00 to 2:00 p.m. ET. The presentation will focus on the initiation of oral difelikefalin Phase ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you