Orthopedic Materials Equipment For Medical Specialties
-
Manufactured by SURJEXbased in PAKISTAN
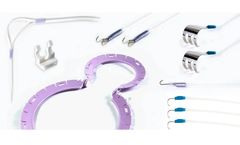
Guide Wire Introducer with Four Pegs S-6304. ...
-
Manufactured by JUNE Medical USA Incbased in USA
Based on our award-winning Galaxy II retractor, the male kit brings the same level of innovation to male ...
-
Manufactured by Morsa Cyberg Orthopedicsbased in TURKEY
It is used for the purpose of traction of head weight with trunk support and restriction of neck movements after fractures, dislocations, ligament injuries in the cervical region and surgical operations of the cervical ...
-
Manufactured by Synimedbased in FRANCE
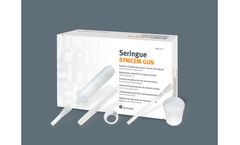
Disposable sterile elements; Contents: Syringe body with plunger. Syringe cover with thread for cannula. Long injection cannula. Short injection cannula. Mixing container. Spatula. ...
-
Manufactured by GerVetUSAbased in USA
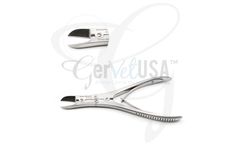
Ruskin Bone Cutting Forceps 6" Curved are double spring, single-action forceps that feature forward or backward angled blades and are commonly used for cutting small ...
-
Manufactured by Bonegraft Biyolojik Malzemeler San. VE TIC. A.S.based in TURKEY
Bone Cement based on polymethyl methacrylate (PMMA) is a widely used biomaterial due to its ease of use in clinical practice and especially the long survival rate proven by dentures. Common Indications for Bone Cement: total joint replacement are bone and joint reconstructions, fracture fixation and treatment of osteoporotic vertebral fractures. Bone Cement consists of two phases, solid and ...
-
Manufactured by Katsan Medical Devicesbased in TURKEY
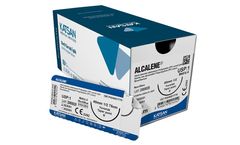
Polypropylene: ALCALENE® sutures are synthetic non-absorbable sterile surgical sutures which are composed of isotactic crystalize polypropylene polymer of high molecular weight. Suture is pigmented blue to enhance visibility. ALCALENE® sutures meet all requirements for synthetic non-absorbable surgical sutures specified at the European Pharmacopoeia (EP) and United States Pharmacopoeia ...
-
Manufactured by Teknimedbased in FRANCE
VACUUKIT® has been designed to contribute to the porosity reduction during the preparation of bone cement by applying vacuum and thereby increase the mechanical stability. The other interests of this technique are the reduction of the MMA fumes to limit exposure of medical ...
-
Manufactured by Greens Surgicals Pvt. Ltd.based in INDIA
Cat. No.: 321.116L; Metal: SS; Sizes: LC-LCP Clavicle Hook Plate - ...
-
Manufactured by CONMED Corporationbased in USA
For surgeons who need strong fixation in a small footprint, the PressFT Suture Anchors combine small size with exceptional strength. The implant comes single or double-loaded with Hi-Fi® suture and is available in PEEK or GENESYS™ biocomposite material. The versatile PressFT™ can be used for various procedures in several joint spaces. ...
-
Manufactured by The Orthopaedic Implant Company (OIC)based in USA
In today’s world of healthcare, surgeon success is being defined as consistent and optimal outcomes underscored by improving value. At OIC, we understand the challenges that arise from a shifting paradigm. That’s why we designed the OIC Anchor to preserve surgeon preference and deliver unparalleled value. The OIC 5.5mm Suture Anchor accommodates up to three number 2 sutures of any ...
-
Manufactured by Mizuho OSIbased in USA
The gold standard for patient positioning in fracture and hip procedures, the Hana is revolutionizing orthopedic ...
-
based in USA
Transet is a fracture fixation system that employs suture and anchors to set bones and tissue. Anchors are applied to the outer bone cortex with suture running across the fracture, providing stabilization and linear fixation of the bone or tissue fragments. This compresses the bone or tissue fragments to promote ...
-
Manufactured by CONMED Corporationbased in USA
With it's small size and innovative instrumentation, the Y-Knot® Flex System was designed to help achieve optimal anchor placement. The anchors provide up to 55% greater fixation strength than conventional 3.0mm press fit anchors while removing up to 80% less bone.1 The curved and percutaneous delivery systems enable delivery around a curve and perpendicular placement for posterior SLAP and ...
-
Manufactured by OSARTIS GmbHbased in GERMANY
BonOs® Inject is a PMMA bone cement that is specifically adapted for spine surgery. The cement is suitable for the fixation and filling of symptomatic vertebral compression fractures, vertebral tumors and vertebral hemangioma as well as for the augmentation of pedicle screws* where bone quality is poor, e.g. in patients with osteoporosis or degenerative or neoplastic ...
-
Manufactured by The Orthopaedic Implant Company (OIC)based in USA
The OIC DRPx Distal Radius Plate represents a best-in-breed approach to wrist fracture and care. As distal radius fractures have become one of the most common injuries treated with internal fixation, OIC supports surgeon and facility in achieving great outcomes under increasing reimbursement pressures. We believe in creating better value for the implants you already know and use on an everyday ...
-
Manufactured by Merete Technologies, Inc. (MTI)based in USA
OsteoBridge™ IDSF is a modular diaphyseal implant used for long term stabilization of segmental bone defects in humerus, femur and tibia of oncology patients. The modular endoprosthesis spacer consists of a spacer(s), that fixates two intramedullary nails and bridges the defect after ...
-
Manufactured by Collagen Matrix, Inc.based in USA
A mechanically strong, compression resistant biocomposite matrix that is easily cut and customized and is fully resorbed during bone remodeling. It is osteoconductive and when mixed with autogenous bone marrow becomes osteoinductive and ...
-
Manufactured by Tecres S.p.A.based in ITALY
Cemex® XL is the low viscosity bone cement without antibiotic. Its low viscosity makes it suitable for syringe application. Available in the 50g pack. Cemex® is the bone cement designed by Tecres for joint implants fixation. Right from the start, the company focused on research and development of acrylic resins, creating a unique and revolutionary product in 1986. Since then, the ...
-
Manufactured by Microbion Corporationbased in USA
Postoperative orthopedic infections, particularly antibiotic-resistant infections, present a serious clinical challenge to surgeons and other treating physicians, since these infections involve surgically installed orthopedic hardware which are frequently associated with persistent microbial biofilms.1 Surgical intervention, irrigation, debridement and potential replacement of orthopedic ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you