Vascular Disease News
-
AngioDynamics Completes Patient Enrollment in APEX-AV Study Assessing AlphaVac F1885 System in Treatment of Pulmonary Embolism
AngioDynamics, Inc. (NASDAQ: ANGO), a leading and transformative medical technology company focused on restoring healthy blood flow in the body’s vascular system, expanding cancer treatment options and improving patient quality of life, today announced the completion of patient enrollment in the Acute Pulmonary Embolism Extraction Trial with the AlphaVac System (APEX-AV). APEX-AV is a ...
-
Bayer receives U.S. FDA Fast Track Designation for asundexian atrial fibrillation program
The OCEANIC-AF (atrial fibrillation) study with asundexian is part of the Phase III OCEANIC clinical trial program enrolling more than 27,000 patients in over 40 countries Asundexian is being evaluated as a potential improved treatment option in stroke prevention and could be part of an entirely new class of treatment options in thrombosis management that aims to uncouple efficacy from ...
By Bayer AG
-
New real world study data on reduced risk of adverse kidney outcomes for Xarelto compared to VKA
In the prospective observational XARENO study, after a two year follow up period, Xarelto was associated with a reduced risk of adverse kidney outcomes and all-cause mortality in patients with non-valvular atrial fibrillation (NVAF) and advanced chronic kidney disease (CKD), compared to vitamin K antagonists (VKA) XARENO evaluated the impact of Xarelto in patients with NVAF and CKD, given that ...
By Bayer AG
-
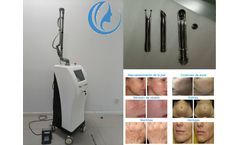
The efficacy of IPL intense pulsed light machine
IPL photorejuvenation machine is the earliest laser skin treatment equipment. It mainly uses IPL intense pulsed light to hit the skin, which has the effect of removing freckles and sealing blood vessels, that is, improving the state of vasodilation. It can also shrink pores, make the skin smoother, and improve the aging state of the skin. Bvlaser is a professional IPL beauty machine factory ...
-
Alfa Chemistry Enriches API Supply by Adding Lenalidomide, Parecoxib Sodium, and Rivaroxaban
As one of the leading chemical manufacturers and suppliers globally, Alfa Chemistry is well-prepared to provide a number of active pharmaceutical ingredients (APIs), such as donepezil and dexmedetomidine hydrochloride, for customers worldwide ranging from lab to commercial scale. Earlier this month, the company announced that three other APIs had been introduced for its global customers as well, ...
-
Soundbite Medical Enters Into A License Agreement With VFLO Medical To Bring Its Products To Greater China
Soundbite Medical Solutions Inc. (SBMS), a medical device company dedicated to developing solutions for the interventional treatment of calcific occlusive peripheral and coronary arterial diseases, today announced that it has entered into an exclusive license agreement with VFLO Medical (VFLO). Pursuant to the agreement, Soundbite has granted to VFLO an exclusive license for certain proprietary ...
-
Bayer receives positive CHMP opinion for Eylea in the EU for treatment of preterm infants with retinopathy of prematurity
Recommendation is based on the Phase III study FIREFLEYE and data from the follow-up study FIREFLEYE NEXT Retinopathy of prematurity (ROP) can lead to severe visual impairment and blindness Bayer will apply for a patent term extension for the patent covering aflibercept, the active ingredient in Eylea™, of six months once the European Commission adopts a decision for a label extension ...
By Bayer AG
-
Prof. Krestin Joins Fluidda’S Board of Directors
INTRODUCTION Gabriel Krestin received his MD from the University of Cologne, Germany where he also specialized in radiology, in 1981 and his PhD (Habilitation) in 1989. From 1990–1997, he worked at the University Hospital in Zürich/CH, including a period as acting chairman of the Department of Radiology. In 1997 he moved to Erasmus MC in Rotterdam, Netherlands where he was the ...
By Fluidda
-
PharmAbcine Announces Poster Presentations on Its anti-VISTA Antibody Candidate at SITC 2022
PharmAbcine Announces Poster Presentations on Its anti-VISTA Antibody Candidate at SITC 2022 DAEJEON, South Korea, October 17, 2022 /PRNewswire/ -- PharmAbcine Inc. (KOSDAQ: 208340ks), a clinical-stage biotech company focusing on the development of antibody therapeutics, announced today that the Company will present preclinical data of PMC-309, one of the Company’s first immuno-oncology ...
-
Stanford Surgeon Venita Chandra Joins Alucent Biomedical Scientific Advisory Board as Company Enters Arteriovenous Fistula Market
Alucent Biomedical, a privately held medical technology company founded to transform the way vascular disease is treated, today announced Venita Chandra, M.D., will join the company’s scientific advisory board. Dr. Chandra is board certified in both general and vascular surgery. She is a clinical associate professor of surgery in the Division of Vascular Surgery at Stanford University ...
-
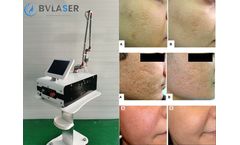
What is CO2 Fractional Laser Treatment
CO2 fractional laser resurfacing machine treatment can quickly improve skin texture, firm skin, improve large pores, and make skin smooth and tender like water. CO2 fractional laser technology is now the world's leading technology for wrinkle removal and skin rejuvenation. Through pulsed fractional laser, laser beauty has been raised to a new level. Bvlaser is a professional CO2 fractional laser ...
-
1st Successful Heart Tumor Removal Without Open Heart Surgery
EchoPixel has once again contributed to a pioneering procedure at CentraCare – St. Cloud Hospital with the first team to remove a heart tumor without invasive open heart surgery using the novel AlphaVac catheter. Previously CentraCare had been the first to use EchoPixel’s 4D hologram technology to conduct the WATCHMAN implant, which is a one-time, minimally invasive procedure ...
-
Inari Medical Announces Results from the Fully Enrolled 800-patient US Cohort of the FlowTriever FLASH Registry
Inari Medical, Inc. (NASDAQ: NARI) (“Inari”) a medical device company focused on developing products to treat and transform the lives of patients suffering from venous and other diseases, announced positive outcomes of the fully enrolled 800-patient FLASH registry in pulmonary embolism (“PE”). The data was presented during a Late-Breaking Clinical Trial session at the 2022 ...
-
Inari Medical Announces Randomized Controlled Trial Evaluating Clinical Outcomes of the ClotTriever System in Deep Vein Thrombosis
Inari Medical, Inc. (NASDAQ: NARI) (“Inari”) a medical device company with a mission to treat and transform the lives of patients suffering from venous and other diseases, announced planned enrollment of the DEFIANCE trial. DEFIANCE is a randomized controlled trial (“RCT”) comparing the clinical outcomes of patients with iliofemoral deep vein thrombosis ...
-
Bayer initiates landmark Phase III study program to investigate oral FXIa inhibitor asundexian
The OCEANIC program will start with two Phase III studies investigating the efficacy and safety of asundexian in prevention of stroke events in patients with atrial fibrillation as well as patients with a non-cardioembolic ischemic stroke or high-risk transient ischemic attack involving up to 30,000 patients Bayer drives the development of its FXIa inhibitor, with the goal of offering a ...
By Bayer AG
-
What is IPL laser?
IPL photorejuvenation treatment is currently the most advanced high-tech beauty program, and it is also the safest and most effective beauty program. Then the theme of price and cost performance that everyone is concerned about, I will give you a comprehensive analysis! Bvlaser Bestview is commercial IPL machine supplier, we have professional IPL machine for sale. What is IPL photorejuvenation? ...
-
CO2 Fractional Laser Scar Removal Equipment In Medical Beauty Industry
With the continuous development of modern medical beauty technology, people's increasingly diversified needs for beauty are also being further met. Especially in recent years, all kinds of medical beauty equipment and technologies have blossomed, and the demand for facial rejuvenation has exploded. The choice of technology will inevitably make it difficult for consumers. Want to have smooth and ...
-
Reva medical announces closing of strategic financing
REVA Medical, LLC. (“REVA” or the “Company”), a leader in bioresorbable polymer technologies for vascular applications, is pleased to announce the closing of a $45 million Series B equity financing. This funding was led by a global strategic investor with deep experience in medical devices, as well as BioStar Capital, and existing investors. Proceeds from the financing ...
-
Robocath’s joint venture, Cathbot, concludes first clinical study in China to evaluate robotic PCI safety and efficacy
End of study brings Robocath closer to commercializing R-One in China in 2023 149 patients enrolled over six months in four Chinese centers Results of first vascular robotics study on this scale in China will be published in March 2023 Robocath, a company that designs, develops and commercializes innovative robotic platforms for the treatment of vascular diseases, announces today that ...
By Robocath
-
£1.1 million project to develop new biodegradable stents
A £1.1 million project to develop biodegradable stents for patients with severe vascular disease, which affects around one million people in the UK, has started thanks to Innovate UK funding. The unique project, a collaboration between the UK-based medical device company Arterius and the Translational Biomedical Research Centre (TBRC) at the University of Bristol, will develop a new type of ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you