Clinical Regulations (Clinical Trials) Equipment
-
Manufactured by Magic Care Health at Home a brand of Flaem Nuova Spabased in ITALY
Easy-to-read large display area, Irregular heartbeat indicator, 60 memories, Scratch-proof soft ...
-
Manufactured by Ito Co., Ltd.based in JAPAN
Electrotherapy Device with 2 Independent Channels and Body Diagram ...
-
Manufactured by Newmars Technologies Ltd.based in HUNGARY
Market-leading needlefree IV connector technology: Help Reduce Infection Risks, Standardize Clinical Protocols, Optimize Supply of IV Consumables ...
-
Manufactured by Ito Co., Ltd.based in JAPAN
Electrotherapy Device with 4 Independent Channels and Body Diagram ...
-
Manufactured by Shree Umiya Surgical Pvt. Ltd.based in INDIA
Medical grade PVC tubing for higher bio-compatibility. Advantage of built-in Heparin and Saline line. Extra corporeal blood circuit tubing set, used during dialysis. Low Priming Volume. Easy of use. Customised blood tubing set as per clinic protocol and for different dialysis machines are available. Injection Site : Large finger guard. ...
-
Manufactured by Bisco, Inc.based in USA
-
Manufactured by Pepex Biomedical, Inc.based in USA
According to the Centers for Disease Control and Prevention (CDC), Sepsis is the tenth most common cause of death overall in the U.S. Sadly, there is no commercially available test that actually detects the sepsis bacteria and current clinical protocols are simply a process of ...
-
Manufactured by Rx Count Corporationbased in USA
Meet the easy, accurate way to automatically count tablets and capsules. For any environment where efficiency matters, you can count on the ...
-
Manufactured by MIS Implants Technologies Ltd.based in ISRAEL
MIS M4 implants combine the benefits of cylindrical and conical implant designs, aiming to achieve high primary stability in any clinical scenario. The two main features of M4 implants are: Self-tapping, V-shaped thread design with three spiral channels, allowing smooth insertion even in type 1 bone. A flat, cutting, tapered apex, ...
-
Manufactured by Tens Unitsbased in USA
The InTENSity CX4 with Therapy Cart is an advanced combination four-channel electrotherapy and ultrasound system offering the practitioner a wide range of treatment options in one user-friendly, ergonomic design. Equipped with a color, touchscreen menu-driven interface intuitively groups and displays clinical protocols, guiding clinicians ...
-
Manufactured by Richmarbased in USA
The TheraTouch EX4 is an advanced four-channel electrotherapy system offering a wide range of treatment options in one user-friendly, ergonomic ...
-
Manufactured by Trina Bioreactives Agbased in SWITZERLAND
Over 50’000 donors for biospecimen from globally different diagnostic and demographic individuals. (Europe, Asia, Americas, Africa) . Different Matrixes (Serum, EDTA, LiHep, etc.) & Multibleeds 1 - 800 ml per Donor. Clinical Data: Patient Data (Age / Gender / GT) Detailed Anamnesis ...
-
Manufactured by Viorabased in USA
Want to meet your customers’ needs with advanced light and RF technology? Viora’s V20 Multi-Technology Platform delivers! Viora’s V20 Multi-Technology platform – part of the V-Series – provides treatment solutions for the most requested ...
-
Manufactured by Inventis SRLbased in ITALY
The Dynamic SPS enables to measure the static and dynamic balance of a patient and to assess the somatosensory, visual and vestibular inputs of a ...
-
Manufactured by Richmarbased in USA
The TheraTouch CX2 and Theratouch CX4 are advanced combination two and four-channel electrotherapy and ultrasound systems. Both offer a wide range of treatment options in one user-friendly, ergonomic design. The CX4 offers four channels for stimulation, while the CX2 is a more compact two-channel ...
-
Manufactured by Bio-Med HealthCare Products Pvt. Ltd.based in INDIA
Medical grade PVC tubing for higher bio-compatibility. Advantage of built-in Heparin and Saline line. Extracorporeal blood circuit tubing set, used during dialysis. Low Priming ...
-
Manufactured by Modus Medical Devicesbased in CANADA
The QUASAR Heavy Duty Respiratory Motion Platform is a programmable breathing and patient motion simulator for the quality assurance of motion-guided radiation therapy systems using any large-scale phantoms up to 45 kg (100 lbs). The Platform HD’s unique multi-directional motion simulation capability enables in-line translation with both the sagittal plane in the SI direction, or at a ...
-
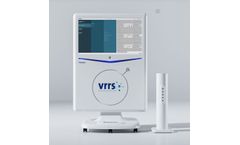
Manufactured by Khymeia Groupbased in ITALY
VRRS Evo is an internationally patented Class I medical device. The most advanced, complete, and clinically proven virtual reality system for rehabilitation and tele-rehabilitation in the world. The extreme ease of use, the vast library of exercises, the innovative sensors, the complete automatic reporting, the high customization ...
-
Manufactured by ACP - Accelerated Care Plus Corporationbased in USA
The OmniSWD® Shortwave Diathermy System provides electromagnetic energy for healing and heating effects in the body. The thermal setting improves treatment efficiency by 25%. With ACP's integrated, evidence-based clinical protocols, on-screen tuning, and a high efficiency output, the OmniSWD saves therapists valuable time to focus on ...
-
based in GERMANY
Restricted uptake of virus particles often lowers the success of gene or shRNA expression and requires the application of higher viral titers. However, increasing the amount of viruses to boost expression can lead to toxic side effects in many cell types and makes the application unnecessarily ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you