In Vivo Models News
-
Showcase
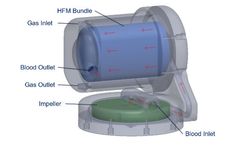
ALung Announces Commercial Development of its Breakthrough Next Generation Artificial Lung
PITTSBURGH–(BUSINESS WIRE)– April 4, 2020 – ALung Technologies, Inc., the leading provider of low-flow extracorporeal carbon dioxide removal (ECCO2R) technologies for treating patients with acute respiratory failure, announced the recent initiation of commercial development of its next generation artificial lung, which expands the Company’s focus on highly efficient gas ...
-
Analyzing Delivery Systems: CD Formulation Introduces In Vitro Testing to Measure Microsphere and Microneedle Release
Researchers and scientists at CD Formulation, a leading pharmaceutical company, have achieved a breakthrough in drug delivery systems with the introduction of an innovative in vitro testing method. This method allows for the precise measurement of release rates for microspheres and microneedles, providing valuable insights for drug formulation and optimizing therapeutic outcomes. By incorporating ...
-
Alfa Chemistry Leads the Way in Tailored Preclinical Drug Research Solutions
Alfa Chemistry serves as an emerging pharmaceutical research organization with a mission to revolutionize the field of preclinical drug research with its wide range of comprehensive services. With a commitment to quality and excellence, Alfa Chemistry offers pharmacological analysis, drug safety evaluation, pharmacokinetic analysis, and bioanalytical services to streamline the drug development ...
-
CD Bioparticles Announces New PLGA-Based Drug Delivery Systems for In Vitro and In Vivo Applications
With years of experience in the pharmaceutical and life science sectors, CD Bioparticles has recently launched its new line of PLGA-based drug delivery systems for in vitro and in vivo applications, including biocompatible and biodegradable PLGA nanoparticles with different surface modifications (e.g., COOH groups, methoxy PEG, and PEG-azide). In recent years, the development of nanotechnology ...
-
Bayer strengthens gene therapy portfolio with lipid nanoparticle technology from Acuitas Therapeutics
Clinically validated lipid nanoparticle (LNP) delivery technology will further strengthen Bayer’s gene therapy portfolio High potency ionizable lipid technology and LNP carriers allow for efficient, targeted, and transient delivery of gene editing RNA components to the liver Bayer AG is joining forces with Acuitas Therapeutics, Inc., a biotechnology company specializing in the ...
By Bayer AG
-
CD Bioparticles Launches mRNA Transfection Reagents for Research Applications
CD Bioparticles, a leading manufacturer and supplier of numerous drug delivery products and services, recently introduced a line of highly efficient mRNA Transfection Reagents with low toxicity for research applications, including the mRNA Transfection Reagent, Stem mRNA Transfection Reagent, and mRNA/gRNA Transfection Reagent. mRNA transfection is the process of introducing and expressing mRNA ...
-
CD Formulation Provides Pharmaceutical Testing on Tablet Fragility, Dissolution, and Disintegration
Pharmaceutical testing is a must to ensure that all medications meet the top quality, safety, and performance requirements before they enter the market. CD Formulation’s cGMP-compliant laboratory is well-equipped to identify both the chemical and structural composition of each drug substance. More recently, the company announced to broaden its service range to tablet fragility test, ...
-
Creative Enzymes Introduces Two Enzyme Engineering Methods to Accelerate Related Research
Creative Enzymes, a professional enzyme provider located in New York, USA, is always hammering away at research and trials in order to provide customers with enzyme services and products with its greatest effort. Just a while ago, the company announced the launch of its reliable enzyme engineering services through site-directed mutagenesis (SDM) and directed evolution, aiming to advance ...
-
Beyond Cancer™ Presents Promising New In Vivo Data Showing Ultra-High Concentration Nitric Oxide Therapy (UNO) in Combination with Anti-PD-1 During the Society for Immunotherapy of Cancer (SITC) 2022 Annual Meeting
Beyond Cancer, Ltd., an affiliate of Beyond Air, Inc. (NASDAQ: XAIR) that is focused on developing ultra-high concentration nitric oxide (UNO) for the treatment of solid tumors, today announced promising new in vivo combination data that support the potential of the company’s novel gaseous nitric oxide (gNO) therapy to treat various types of solid tumors in combination with immune ...
-
ViGeneron presents preclinical data on intravitreal gene therapy of Stargardt disease at ESGCT
Efficient transfer of larger gene via ViGeneron’s proprietary technology platforms REVeRT and vgAAV ViGeneron advances its preclinical programs for Stargardt disease and Retinitis Pigmentosa into IND-enabling activities and clinical stage development ViGeneron GmbH, a next-generation gene therapy company, today announced the presentation of preclinical data on its program VG801 in ...
-
Lumen Bioscience Awarded $3.6 Million by U.S. Army to Advance Low-cost, Scalable Preventative for Antibiotic-Resistant Infections
Lumen Bioscience—a clinical-stage biopharmaceutical company developing biologic drugs for highly prevalent diseases—today announced the receipt of a $3.6 million award from the Department of Defense (DoD) for advanced biomedical product development in support of Naval operations. The award from the Medical Technology Consortium (MTEC), a 501(c)(3) biomedical technology consortium ...
-
Oligomerix Presents Data from IND-Enabling Studies on Clinical Candidate at Clinical Trials on Alzheimer’s Disease (CTAD) and Neuroscience 2022 Meetings
Oligomerix, Inc., a privately held company pioneering the development of small molecule therapeutics targeting tau for rare neurodegenerative and Alzheimer’s diseases, today announced the presentation of IND-enabling preclinical data on its lead product candidate OLX-07010 at the annual Clinical Trials on Alzheimer's Disease (CTAD) and Society for Neuroscience’s Neuroscience meetings ...
-
Novel Modulators of LDL Metabolism
DESCRIPTION provided by applicant Heart disease is the leading cause of death for both men and women in the US accounting for nearly of all annual deaths A high cholesterol level is well known risk factors for heart disease Although blood cholesterol can be lowered using a number of marketed drugs of which statins are the leading drugs only of patients taking these drugs are achieving the low ...
-
Elpiscience Announces Studies Presented at SITC 2022 Annual Meeting
Elpiscience Biopharmaceuticals, Inc. (“Elpiscience”), a clinical-stage biopharmaceutical company focused on developing next-generation immunotherapies to benefit cancer patients worldwide, presented positive studies for its innovative immunotherapeutic molecules at the SITC 2022 Annual Meeting, including anti-SIRPα monoclonal antibody ES004, PD-L1/SIRPα bispecific antibody ...
By Elpiscience
-
InMed Pharmaceuticals Advances Neurodegenerative Disease Program with Natural Sciences and Engineering Research Council of Canada (`NSERC`) Alliance Grant Funding
Targeting neurodegenerative diseases such as Alzheimer’s, Huntington’s and Parkinson’s Identified two lead cannabinoid analog compounds for in vivo studies NSERC grant funding supports InMed’s collaborative research projects with Dr. Kumar from University of British Columbia (UBC) VANCOUVER, British Columbia, Nov. 16, 2022 (GLOBE NEWSWIRE) -- InMed Pharmaceuticals ...
-
Full-Scale PROTAC In Vivo Evaluation Services to Facilitate PROTAC Drug R&D
In the process of drug development, the evaluation of drug effectiveness is one of the keys to determining whether prodrugs can sufficiently reach the market stage. BOC Sciences, as a professional contract research organization, can take care of the PROTAC in vivo evaluation workload for clients with a transparent project process and comprehensive evaluation reports to facilitate their PROTAC ...
By BOC Sciences
-
Galapagos presents roadmap for pipeline and commercial growth at its R&D Day 2022
Galapagos NV (Euronext & NASDAQ: GLPG) today held its R&D Day 2022, featuring presentations of key opinion leaders and company management on the strategic, scientific, and commercial progress at the company. The company also presented its financial results for the third quarter of 2022. Paul Stoffels3, CEO and Chairman of the Board of Directors of Galapagos, commented: “Today we ...
By Galapagos NV
-
Antengene Receives U.S. FDA Clearance of IND Application for Phase I Trial of Small Molecule ERK1/2 Inhibitor ATG-017 in Patients with Advanced Solid Tumors
ATG-017 is a small molecule ERK1/2 inhibitor and Antengene has obtained exclusive global rights to develop, commercialize and manufacture ATG-017. IND clearance enables Antengene to initiate the combination portion of the Phase I “ERASER” clinical trial in the United States (U.S.) to evaluate the safety, pharmacokinetics, and preliminary efficacy of ATG-017 combination therapy ...
-
CD Bioparticles Launches NIR-Quantum Dots for Biomedical Imaging
With years of experience in the pharmaceutical and life science sector, CD Bioparticles launched a range of near-infra-red (NIR) quantum dots that exhibit excellent optical properties and biocompatibility for biomedical imaging. Currently, CD Bioparticles has successfully synthesized a variety of NIR quantum dots, including PbS/CdS, PbS, PbSe, Ag2S, CuInS/ZnS, and CdTe/CdSe/ZnS. Quantum points ...
-
PharmAbcine Announces Poster Presentations on Its anti-VISTA Antibody Candidate at SITC 2022
PharmAbcine Announces Poster Presentations on Its anti-VISTA Antibody Candidate at SITC 2022 DAEJEON, South Korea, October 17, 2022 /PRNewswire/ -- PharmAbcine Inc. (KOSDAQ: 208340ks), a clinical-stage biotech company focusing on the development of antibody therapeutics, announced today that the Company will present preclinical data of PMC-309, one of the Company’s first immuno-oncology ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you