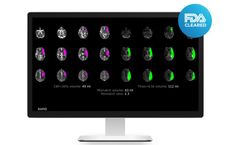
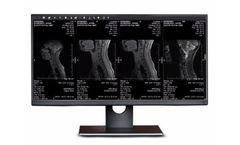
Pre-Clinical Software
-
Manufactured by Intelligencebased in GERMANY
Innoplexus recognizes the unique requirements of the life sciences industry and offers custom solutions on top of our products to enable pharmaceutical and biotech companies as well as CROs to have timely and easy access to useful insights. Our proprietary AI technology combined with a self-learning life sciences ontology that understands the context of the search query and our capability to ...
-
based in USA
The Seven Bridges Platform is a cloud-based environment for conducting bioinformatic analyses. It is a central hub for teams to store, analyze, and jointly interpret their bioinformatic data. The Platform co-locates analysis pipelines alongside the largest genomic datasets to optimize processing. It allocates storage and compute resources on demand, to meet the needs of ever-growing ...
-
Manufactured by Verilybased in USA
Onduo has built a virtual care model that leverages connected devices, advanced software, deep analytics and specialty clinical care to provide lifestyle and clinical interventions with a goal of helping people with chronic conditions live healthier lives. Launched in 2016 with an initial focus on type 2 diabetes, Onduo works closely with parent company Verily to create a next generation platform ...
-
based in USA
Our scientists use the nference platform and data in collaboration with your teams to tackle your most important questions and challenges. nference is creating the largest labeled EMR dataset in healthcare. EMR data contains longitudinal real-world, “deep data” rich in clinical phenotypes and outcomes spanning across therapeutic areas, but exists in in largely semi-structured and ...
-
based in GERMANY
Reliable predictions to anticipate risks. Interlinked data to find new targets and design novel drugs. Real-world evidence to evaluate success. These capabilities eliminate the uncertainties that limit drug development but are data-intensive and experimentally inaccessible. Molecular Health Dataome Technology offers powerful solutions to transform drug development. By merging world-class ...
-
by Lifebitbased in UNITED KINGDOM
The velocity and volume of real world data (RWD) means it is impossible for humans alone to gain ultimate value from this type of information. Lifebit REAL uses Artificial Intelligence to make sense of RWD. Using REAL, you can unlock meaningful insights in real-time, so you can stay at the forefront of science innovation and ...
-
by Formedixbased in UNITED KINGDOM
ryze clinical metadata repository is the easiest way to manage & reuse standards and studies. Imagine if all your study content – standards, forms, datasets, mappings… were in 1 place. And they were all interconnected. Everything from eCRFs to submission deliverables. No matter where they were, your teams could manage standards and studies, share and reuse content – even ...
-
based in RUSSIA
Randomization In Treatment Assignment Based On Aims Of Trial; Randomization in 10 secs. Different types of randomization: simple, block or based on stratification factors. Integration with MATRIX EDC and Trial Supply module. Notifications about subject status for the team. Roles and access levels for trial ...
-
based in GERMANY
Rapidly generate real-world evidence of effectiveness with our modular infrastructure for continuous collection of real-world data. Our RWE generation infrastructure bridges the gap between clinical trials and the real world. Get the right data at the right time while creating tangible value for hospitals and ...
-
by RapidAIbased in USA
Rapid CTP is the only clinically validated software with an FDA indication to aid in the selection of patients for acute stroke ...
-
by Cytelbased in USA
The complexity of conducting regression analysis over multiple covariates is well-documented. The challenge only intensifies when coupled with small sample sizes or missing data sets. LogXact aims to provide simple and accurate solutions for such ...
-
based in USA
For clinicians, Eviti Advisor grants access to Eviti’s vast evidence-based treatment library, providing thorough details on the effectiveness, side effects, and cost of potential cancer treatments. Cancer research is ever-evolving. Often, physicians struggle to manage overwhelming amounts of information around the risks, benefits, and trade-offs of different treatment options. Time is of ...
-
Manufactured by Synaptive Medicalbased in CANADA
Simplify research data management for large-scale medical research initiatives. Automatically collect, de-identify, organize, and share data among research collaborators to enable large-scale clinical ...
-
by Immunaibased in USA
Immunai’s end-to-end single-cell platform enables high resolution profiling of the immune system at industrial scale, generating biological insights that accelerate R&D and power novel ...
-
by Evidationbased in USA
Create new measures of health. Reveal critical between care data. Understand the full patient journey. Evidation’s suite of products allow you to engage with individuals on a whole new level in order to deliver better health ...
-
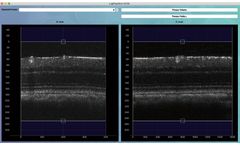
Manufactured by LighTopTech Corpbased in USA
Optical coherence tomography angiography (OCTA) is an invaluable diagnostic technology for non-invasive, label-free vascular imaging in vivo. However, since it relies on detecting moving cells from consecutive scans, high-resolution OCTA is susceptible to tissue motion. LighTopTech developed a novel OCTA technique named ultrahigh-resolution factor angiography (URFA) by ...
-
based in USA
The ASAPprime software is the only commercially-available software that enables pharmaceutical scientists to quickly and accurately determine drug substance and drug product shelf-life based on the Accelerated Stability Assessment Program (ASAP). This state-of-the-art, statistical software allows prediction of small-molecule drug product use periods, shelf-lives and drug substance retest periods ...
-
based in FRANCE
Moovcare® is a digital therapeutic that uses a simple weekly questionnaire to detect relapse or complication during follow-up of lung cancer patients. A Class I medical device, Moovcare has been proven in clinical trials to improve overall survival by 7.6 ...
-
based in USA
Assess the probability of technical and regulatory success (PTRS) and phase transition probability of your pipeline programs in an objective and efficient way. Understand the supporting rationale, track the evolution of PTRS based on key events, and make portfolio and program-level decisions. ...
-
based in USA
Serving the needs of the entire healthcare institution while providing separate interfaces optimized for each of the two workflows. ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you