- Home
- Companies
- Ankasa Regenerative Therapeutics
- Products
- Ankasa - Model ART352-L - Regenerative ...
Ankasa - Model ART352-L -Regenerative Medicine for Better Bone Formation
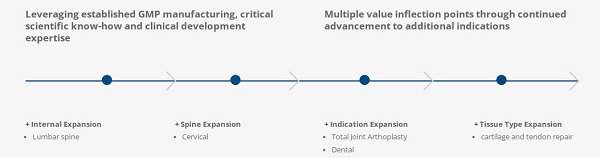
Stem cell renewal and maintenance and tissue regeneration are critical for normal health and well-being. Wnt pathway interactions are crucial to maintenance of these processes. Ankasa is the first to produce human Wnt proteins in a manufacturing setting suitable for use in humans. Our first clinical approach will be the demonstration of improved outcomes after spinal fusion surgeries. Our product has expansion opportunities to other bone reconstructive procedures including osseointegration of dental and orthopedic implants (knee and hip replacements), fracture repair, total joint arthroplasty revisions, and others.
- Large Growing Market(s)
- Novel Product with Compelling Data
- Robust Pipeline Potential
- De-Risked Phase 2 Opportunity
- Proven Executive Leadership
Spinal fusion: $5B+ US/$3B ex-US market
Total joint arthroplasty (hip/knee): $9.5B US market
Disruptive, clinical stage biologic for safe bone healing enchancement
- Liposomally delivered rhWnt3 protein activates resident stem cells to osteogenic phenotype.
- Potential for superior bone formation vs. autograft
Spinal fusion for degenerative lumbar spine disorders as initial indication (IND Q1 ’20)
- Goal is more rapid fusion, higher fusion rates and durability
Scientifically proven platform for pipeline expansion to arthroplasty, fracture and dental applications
GMP manufacturing complete
Unmet medical need: Autograft efficacy declines with age.
In patients >50, autograft becomes unreliable. This functional decline can be attributed to aging skeletal stem cells & insufficient growth factors.
Ankasa’s proprietary Wnt3 protein therapeutic activates skeletal stem cells and accelerates bone regeneration in animal models of spinal fusion, segmental defects and osseointegration.