
AstroRx - Functional Healthy Astrocytes
Clinical grade human astrocytes (the nervous system supporting cells) for the treatment of ALS and other neurodegenerative diseases. Received approval by the Israeli Ministry of Health to commence our phase I/IIa clinical trial in ALS patients. 6 month follow up results for Cohort B have been reported.
Functional Healthy Astrocytes to Protect ALS-Diseased Motor Neurons
ALS is a rapidly progressive and fatal neurological disease, causing death on average within 3-5 years from time of diagnosis. The disease attacks motor neurons responsible for controlling muscles and leads to paralysis and impaired breathing and swallowing. Multiple studies have demonstrated that astrocytes play a crucial role in ALS disease progression. In ALS patients, the astrocytes malfunction and attack the neurons.
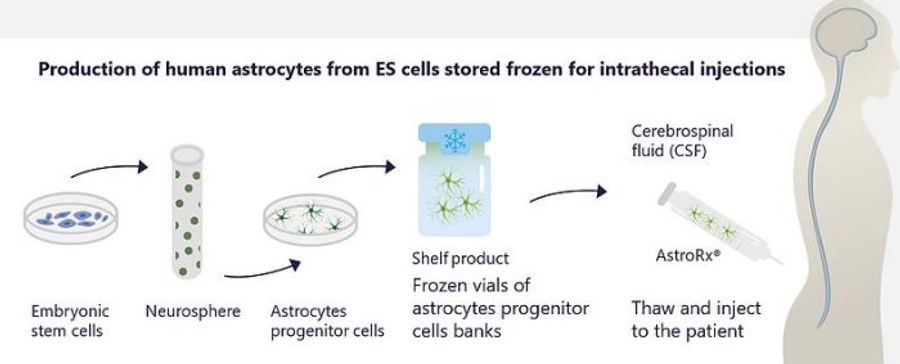
Kadimastem's innovative approach is to use fully differentiated astrocytes as a regenerative medicine therapy for ALS – AstroRx® to slow the progression of the disease and improve the patient's life quality and life expectancy. AstroRx® is administered directly into the patient’s spinal cord fluid by a standard intrathecal injection under local anesthesia. Pre-clinical studies in ALS animal models showed that AstroRx® is a safe treatment that provides systemic support to damaged motor neurons, increasing the life expectancy and motor functions.
More about Functional Healthy Astrocytes
“Most cell-based therapies are likely to be ...
“Most cell-based therapies are likely to be autologous, where stem cells are harvested from a specific tissue of a patient and cultured to create large quantities over many weeks and then returned to the same patient.
Moreover, the cells had been taken from a patient that had already manifested the disease. Another major hurdle of an autologous solution is the need for a lengthy procedure for each patient and the inability to create a ready-to-use product.
We are focused on our two main cell therapy products we invest all our efforts to bringing “off-the-shelf” products to the clinic in the best quality. We accumulated significant knowledge on cell growth, scalability, freezing methodologies, cell characterization, and we are open to new collaborations that can help improve and progress the regenerative medicine field.
One key issue is getting them from the bench to the bedside without compromising safety and efficacy. The successful commercialization of cellular therapeutics requires extending the shelf-life of these therapies through a developmental process called cryopreserved. We are moving fast with this process with AstroRx® and IsletRx. This development will allow banking of large quantities of cell products for broader distribution supporting the next advanced clinical trials and marketing phase.”
Dr. Michal Izrael, PhD
Clinical Trial on ALS Patients using our Innovative Cell Therapy
The Company conducted a phase 1/2a clinical trial on ALS patients
(https/iclinicaltrials.goylct2/showiNCT03482050)
The trial was conducted at Hadassah Ein Kerem Hospital in Jerusalem a world leading center in the field of ALS
Study Design:
- The trial included 10 ALS patients
- The Company has completed treatment (cell transplantation of AstroRx®) in 10 patients of Cohort A and B
- The company has received approval from the MOH of Israel to commence our phase I/IIa clinical trial in ALS patients
- Results of Cohort A and interim results for the Cohort B have been published on our press release page.
- 6 Mo. results for the Cohort B were published in December of 2020
'The results of the clinical trial demonstrated a high safety profile of AstroRx® cells. In addition, the results showed clinical meaningful decline in disease progression as measured by ALS Functional Rating Scale-Revised (ALSFRS-R) during the first 3 months after AstroRx® intrathecal transplantation in both tested doses of AstroRx® cells. We are now moving forward with all the activities needed to test multiple injections of AstroRx cells in our next clinical trials.” Michal Izrael PhD.
Customer reviews
No reviews were found for AstroRx - Functional Healthy Astrocytes. Be the first to review!


