BlueWillow - Vaccine Pipeline
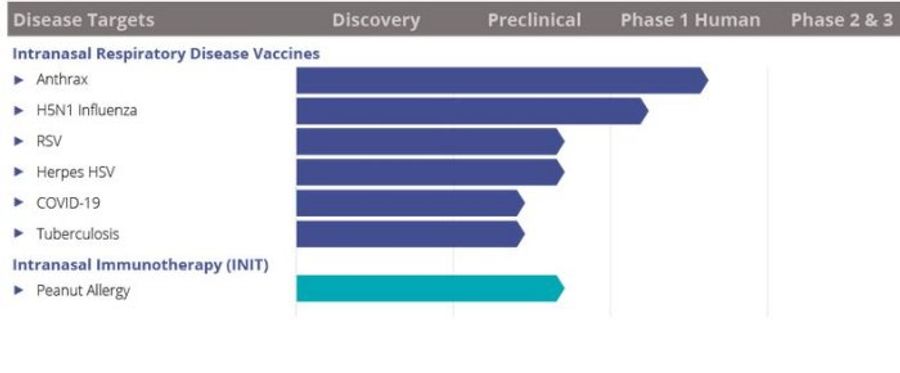
BlueWillow’s intranasal vaccine technology has demonstrated safety and immunogenicity in Phase 1 clinical studies using a seasonal influenza vaccine. Several other ongoing BlueWillow programs — including intranasal vaccines for anthrax, pandemic influenza, RSV, HSV-2 and chlamydia — have recently advanced through successful challenge studies in the respective primary animal model. Click on a program below for details on our research results and partnering information.
-
Most popular related searches
Customer reviews
No reviews were found for BlueWillow - Vaccine Pipeline. Be the first to review!