BrainScope Company, Inc.

BrainScope - FDA Cleared Non-Invasive Medical Device
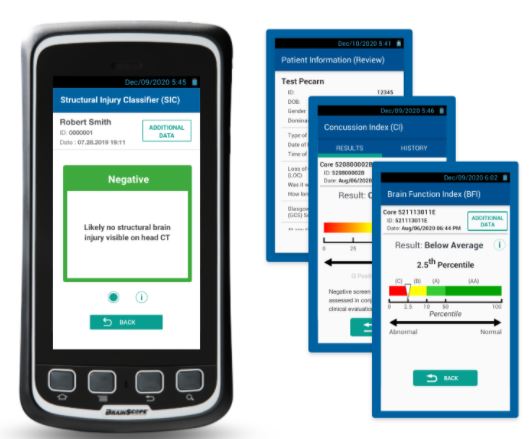
BrainScope is the only FDA cleared non-invasive medical device that objectively assesses head injured patients for both brain bleeds & concussions at point of care.
Most popular related searches
non-invasive medical device
medical device
brain bleed
clinical validation
brain injury
brain concussion
brain activity
- 8 FDA clearances
- 12 years of R&D
- 32 peer reviewed publications
- 13,000+ subject evaluations
Designed for accurate, fast & easy assessment of suspected mTBIimage-png-Feb-09-2021-03-09-15-53-AM
Accurate
- Confirm the likelihood of a brain bleed with 99% sensitivity to the smallest detectable level of blood (>1 mL).
- Determine concussion status at same time with same setup and recording.
Fast
- Significantly reduce patient length of stay in the emergency department.
- Prep, test, result in under 20 minutes.
Easy
- Integrate easily into busy,
- infection-prone environments.
- Use the disposable headset on masked patients and disinfect device with wipes.
FDA cleared A.I. derived algorithms assess 10,000 EEG features and employ age regression and advanced pattern recognition to determine the likelihood of structural and functional brain injury with high independently validated accuracy.