

- Home
- Companies
- Biocardia, Inc.
- Products
- CardiAMP - Cell Therapy System
CardiAMP - Cell Therapy System
The investigational CardiAMP™ Cell Therapy is designed to be a comprehensive biotherapeutic heart failure solution, incorporating: a proprietary molecular diagnostic to characterize the potency of a patient’s own bone marrow cells and determine if they are an optimal candidate for therapy. a point of care processing platform to prepare cells at the patient’s bedside. an optimized therapeutic formulation that builds on the total experience in the cardiac stem cell field to-date. a proprietary interventional delivery system that easily navigates a patient’s vasculature to securely deliver the specific dosage of cells in a routine cardiac catheterization procedure.
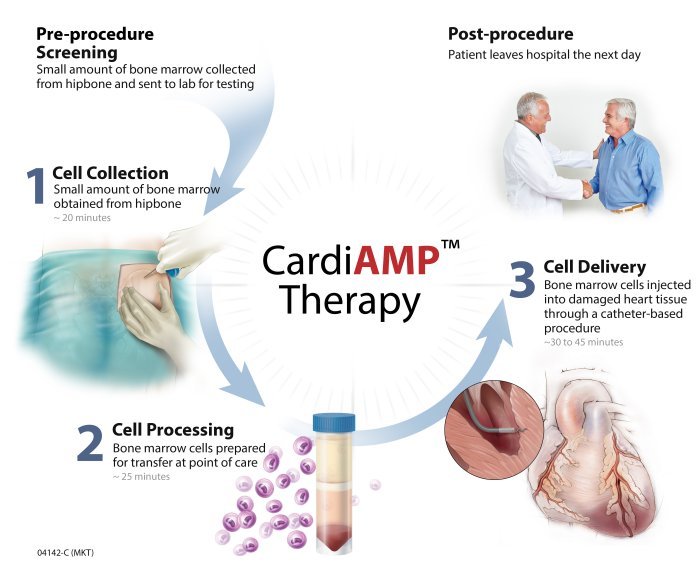
Day 1: Mini bone marrow aspirate (~ 1 tablespoon) of patient’s bone marrow cells from their hip bone. Aspirate sent to BioCardia CLIA laboratory partner. BioCardia’s proprietary molecular diagnostic assay identifies patients with potent cell characteristics using an In Vitro Diagnostic Multi Indexed Assay.
Day 3 or after: If assay positive, patient presents for 1-hour treatment in cardiac catheterization lab:
- Small bone marrow aspirate ( ~4 tablespoons) ~15 minutes
- Cells minimally processed and concentrated into proprietary dosage
- Cells delivered using proprietary Helix Transendocardial Delivery System
- Patient discharged same day or after overnight stay
In both the Phase I clinical trial of 20 patients (the TABMMI trial) and Phase II randomized placebo-controlled trial of 30 patients (the TACHFT-BMC Trial) CardiAMP cells demonstrated an excellent safety profile in heart failure patients treated at two dosages. There have been no incidences of treatment-emergent major adverse cardiac events. The Phase II results from TACHFT-BMC support efficacy relative to patient quality of life and functional capacity, which was shown to be both statistically and clinically significant. The FDA has approved us to begin enrollment of patients in our CardiAMP Phase 3 pivotal, pre-commercial trial using a patient`s own cells, administered in the cardiac catheterization lab directly into the heart muscle in cases of serious heart failure following a prior heart attack. The trial is expected to begin in approved centers in late 2016 or early 2017.
About 5.7 million Americans are living with heart failure today, with about half a million more diagnosed each year. The CardiAMP Heart Failure Trial is studying an investigational stem cell-based therapy for heart failure that develops after a heart attack. The therapy takes a personalized and minimally-invasive approach to the use
of a patient’s own cells in the treatment of ischemic heart failure, and is designed to stimulate the body’s natural healing response.
The CardiAMP heart failure study is enrolling up to 260 patients at 40 medical centers throughout the United States. The study participants will be randomly placed into one of two groups, a treatment and a sham control group. Neither the patient nor the researcher will know who will be receiving the CardiAMP cell therapy and who will be receiving the sham treatment. Both groups will be closely monitored throughout the study by a team of physicians and continue to be treated with standard of care medical treatments.
Transplanted cells are hypothesized to benefit the heart through direct and indirect pathways, accelerating the body’s natural healing process. We do not believe these cells transform into cardiomyocytes or heart cells.
- Direct Regeneration: Transplanted cells actively home to injury sites and differentiate into new functional tissue to augment organ function.
- Indirect Regeneration: Transplanted cells secrete stimulatory cytokines to instigate an innate regenerative response from resident stem cells. Cytokines are cell signaling molecules that aid cell to cell communication in immune responses and stimulate the movement of healthy cells towards sites of inflammation, infection and trauma.
