

- Home
- Companies
- Nanostics Inc.
- Products
- ClarityDX Prostate - Prostate Cancer ...

ClarityDX Prostate - Prostate Cancer Blood Test Kit
Our lead product, ClarityDx Prostate® is designed to be a highly accurate test to assist in diagnosing clinically significant prostate cancer and improve prostate healthcare decisions. Validation of ClarityDX Prostate® is ongoing in a 2800 patient clinical study designed to meet regulatory approval.
When ClarityDX Prostate® comes to market, it will help physicians and patients navigate biopsy decision at TWO critical points in care
Initial diagnosis:
ClarityDX Prostate® is designed to accurately predict the presence of clinically significant prostate cancer and with the guidance of a physician can be used to inform the decision to biopsy.
Active surveillance:
ClarityDX Prostate® can be used to monitor patients on active surveillance to detect the progression to clinically significant prostate cancer and inform the decision to biopsy.
The ClarityDX Prostate® test was validated in two prospective clinical trials in partnership with APCaRI and DynaLIFE.
The test achieved accuracy measurements of > 0.8 AUC (Area Under the Curve) for the identification of clinically significant prostate cancer, compared to 0.58 AUC for the current standard of care Prostate-Specific Antigen (PSA) test.
Our results showed a 40% increase in accuracy over PSA testing.
Sector
Diagnostics and Liquid Biopsy.
Platform
Developed with the ClarityDX® Extracellular Vesicle Machine Learning Analysis Platform.
Clinical Trials
AUC: > 0.8
Sensitivity: 98%
NPV: 98%
Our test detects clinically significant prostate cancer with the highest accuracy in prospective clinical trials compared to similar tests in the market.
Successful implementation of ClarityDX Prostate® could eliminate up to 600,000 unnecessary biopsies and 24,000 related hospitalizations in North America each year.
By differentiating between indolent and clinically significant prostate cancer, ClarityDX Prostate® can guide physicians and patients through the decision process. As a result, men may be spared the adverse events of biopsy, prostate cancer surgery, radiotherapy, and hormone therapy.
When ClarityDX Prostate® gets to the market the test will save an estimated $1.4 billion in healthcare costs by reducing the number of prostate biopsies and unnecessary treatments such as surgery and radiation.
ClarityDX Prostate® is designed to be a non-invasive and easily performed test with a small equipment footprint, these features will make this test ideal for large-scale population-based screening in medical clinics, worldwide.
ClarityDX Prostate® is under development and so currently has not received regulatory approval or clearance in any markets. ClinicalTrials.gov Identifier: NCT03957252
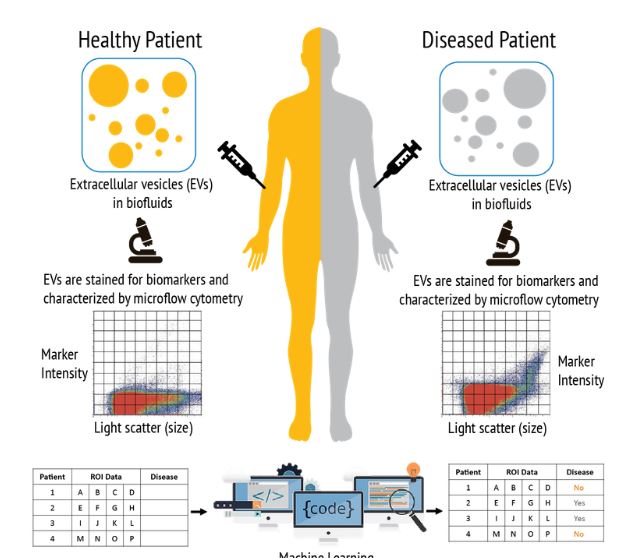
ClarityDX® technology can be used to predict disease The diagnosis and management of chronic diseases is a significant challenge and burden to healthcare systems globally
EVs carrying disease-specific biomarkers like nucleic acid and proteins are continuously released from cells and can be found in biological fluids including blood, urine, semen, and cerebrospinal fluid. The levels of disease-specific EVs are closely related to disease progression making EVs promising targets for minimally invasive diagnostic assays.
Technology designed to accurately detect and measure EVs has the potential to greatly improve liquid biopsy diagnostics.
At Nanostics, we use micro-flow cytometry (µFCM) to quantify EV size, concentration, and marker abundance for millions of EVs in minutes. We then use advanced machine learning to analyze the vast amount of data generated using µFCM to provide rapid and precise results that continuously improve with every test.
The EV and machine learning platform ClarityDX® is set to transform the diagnostic landscape and make easy-to-use, minimally invasive predictive tests a reality in the near future.
