

DTR Medical Ltd, part of the Innovia Medical Group
- Home
- Companies
- DTR Medical Ltd, part of the Innovia ...
- Products
- DTR Medical - Model SNAP - Nasendoscopy
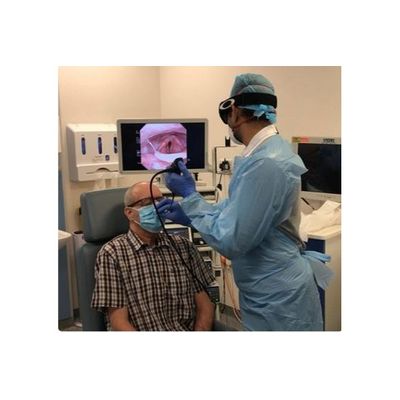
DTR Medical - Model SNAP -Nasendoscopy
Protects from airborne risks and diseases. A one-way valve that can be retrofitted to any fluid resistant Surgical Mask. Designed with the aim to keep Clinicians safe and so better able to deliver high quality care to their patients. Facilitates the movement of a Nasendoscope up to 4mm in diameter. Ensures safe passage of a Nasendoscopy through a Surgical Mask, creating an aperture for the Endoscope to pass through into the Nasal Cavity.
Most popular related searches
surgical mask
COVID 19
nasal cavity
nasendoscopy
surgical
patient movement
endoscopy
disease risk
surgical fluid
- Reduces the risks associated with the patient sneezing
- Scientifically proven to reduce the prevalence of AGP (Aerosol Generating Particles) during examination
- Provides better safety and care for both the Patient & Clinician
- Clinical Otolaryngology Peer Review Paper and Trial
- Filtrex Optical Particle Report for SNAP
- MET 1952-2591 Splash Resistance Test Report
- SNAP White Paper August 2020
- UK Manufactured
- Winner of the 2020 MediWales “Partnership with the NHS” Award
- Winner of the 2021 “Special Focus On COVID” Meridian Award
- Continuing ENT Diagnosis Throughout COVID-19 Safely webinar
- UK Patent Application Number 2105626.2
- Exclusively distributed by DTR Medical
