

- Home
- Companies
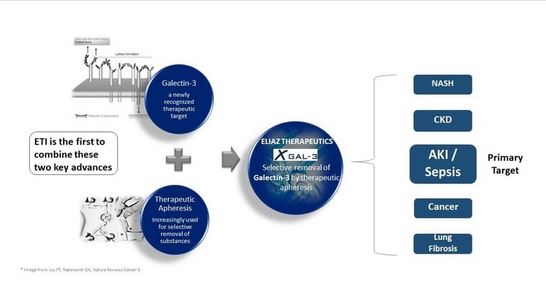
- Eliaz Therapeutics
- Products
- Galectin - Model X-Gal-3 - Acute Kidney ...

Galectin - Model X-Gal-3 -Acute Kidney Injury (AKI) Device
Acute Kidney Injury (AKI) and sepsis are major public health concerns, with high incidence, mortality and morbidity. Despite efforts to develop new interventions, current therapeutic options are limited in scope and effectiveness. At Eliaz Therapeutics, we have developed a patented, unique medical device that will remove Galectin-3 (Gal-3) from human blood. Our device removes both bound and free Gal-3 from the circulation, offering a new treatment option that is less invasive and has fewer side-effects than other therapies.
The device promises to address a significant unmet need for effective interventions in the treatment of AKI and sepsis, in addition to other serious conditions.
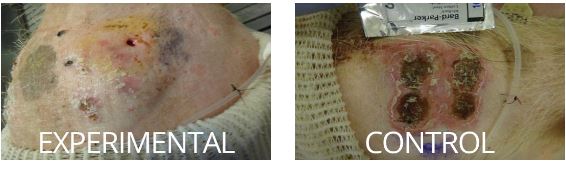
Proof of Concept
Our Proof of Concept was a large animal study that showed how our medical device remarkably reduced inflammation and fibrosis. See the control and experimental photos below.
Acute Kidney Injury (AKI) can be life-threatening in various patient populations.
Full recovery of kidney function is uncommon and leaves AKI patients at risk of long-term morbidity and death.
AKI affects almost 900,000 people in the United States with an annual cost of up to $24.0 billion. The most expensive patients are those with AKI of sufficient severity to require dialysis, where cost increases relative to patients without AKI and can reach up to $42,000 per hospitalization event.
In-hospital, mortality for patients with AKI has recently been estimated between 20 and 25%, while critically ill patients with dialysis-requiring AKI experience mortality rates in excess of 50%. Despite efforts to develop new therapeutic modalities, current therapeutic options are very limited. Global, regional, and national sepsis incidence and mortality, 1990–2017: Analysis for the global burden of disease study.

