

- Home
- Companies
- Adagio Medical, Inc.
- Products
- iCLAS - Ultra-Low Temperature ...

iCLAS - Ultra-Low Temperature Cryoablation (ULTC) System
iCLAS ultra-low temperature cryoablation (ULTC) system has been developed with the vision of bringing the durability of Cox-Maze procedure1 to percutaneous transcatheter treatment of atrial arrhythmias. ULTC technology utilizes -196°C nitrogen flow to rapidly freeze myocardial tissue to form durable, contiguous, transmural lesions2. The data from multi-center European trial of ULTC showed 85% single-procedure freedom from AF at 12 months in persistent AF patients along with 1.5% rate of transient device-related complications3. In the U.S.A. iCLAS system is an investigational device, limited by Federal law to investigational use (IDE #G180623).
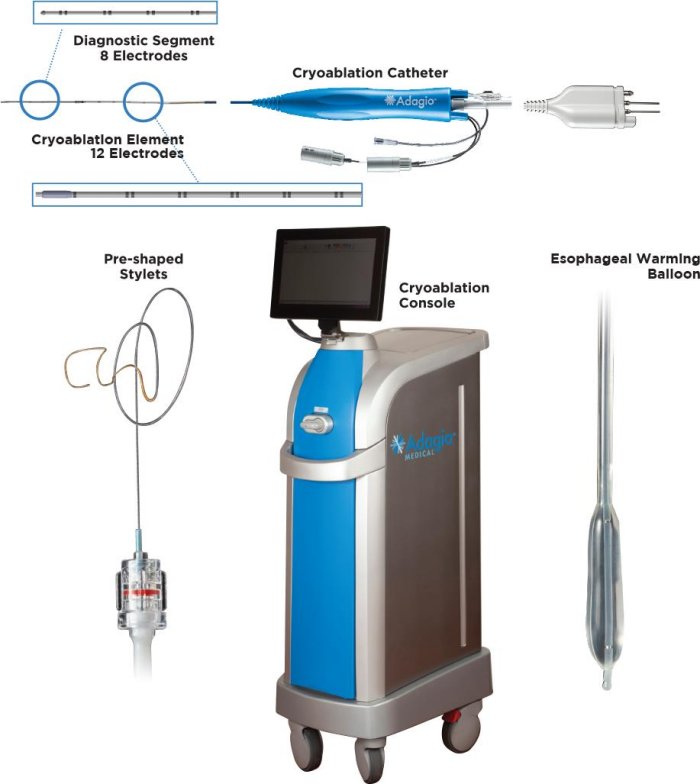
Adagio`s portfolio is based on ultra-low temperature cryoablation (ULTC) designed to produce durable, contiguous, transmural lesions anywhere in the heart, by itself, or with the added benefit of cardiac electroporation (Pulsed-Field Cryoablation or PFCA).
Transcatheter ultra-low temperature cryoablation (ULTC) is Adagio’s Medical proprietary technology. As compared to other commercially-available catheter cryoablation technologies which utilize N2O refrigerant at Tb = -88.50C and rely on slow cell dehydration for lesion formation, ULTC utilizes “near-critical” N2 flow at Tb=-1960C to rapidly cool target heart cells to temperatures consistent with formation of intracellular ice and almost instantaneous death. Unique feature of ULTC technology is it’s ability to produce broad variety of contiguous transmural lesions in both atria and ventricles using a single ablation cycle.
Adagio Medical iCLAS™ Ultra-Low Temperature Cryoablation System is CE-Mark approved for treatment of atrial flutter, paroxysmal and persistent atrial fibrillation, and is an Investigational Device in the USA. The use of ULTC for treatment of monomorphic ventricular tachycardia is being evaluated in the early-stage clinical trial.
First-in-human cases of Adagio Medical’s PFCA for treatment of atrial fibrillation took place in December 2021 at Medicover Hospital in Warsaw, Poland. The use of PFCA for treatment of persistent atrial fibrillation is being evaluated in the early-stage clinical trials.
