Clinical Data Analysis Equipment Available In Canada
-
based in USA
All medical devices sold in the EU must have a Clinical Evaluation Report (CER) as part of its technical documentation to satisfy the new EU Medical Device Regulation (MDR) requirements. A comprehensive analysis of pre- and post-market data relevant to a medical device, a Clinical Evaluation Report demonstrates that your device ...
-
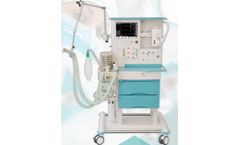
Manufactured by heyer medical AGbased in GERMANY
PASITHEC II is an advanced yet easy to use anesthesia workstation that provides accurate, pneumatically driven and electronically controlled ventilation. It has a user-friendly design, incorporates new technology and provides safe and effective treatment options for the clinician. PASITHEC II includes adult and child modes that provide patient-appropriate defaults and ranges. ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you