Clinically Evaluated Equipment
-
based in USA
All medical devices sold in the EU must have a Clinical Evaluation Report (CER) as part of its technical documentation to satisfy the new EU Medical Device Regulation (MDR) requirements. A comprehensive analysis of pre- and post-market data relevant to a medical device, a Clinical Evaluation Report demonstrates that your device ...
-
Manufactured by Rebiotix Inc., a Ferring Companybased in USA
Rebiotix Inc., a Ferring Company, has leveraged its clinical, manufacturing and regulatory experience with RBX2660 to develop the first of its kind non-frozen, room temperature stable oral microbiota-based formulation under the MRT™ drug platform, known as RBX7455. While it is currently used to reduce the incidence of ...
-
Manufactured by Canfield Scientific, Inc.based in USA
The Standard in Facial Imaging. Strengthen your claims with validated imaging tools. Now includes PRIMOS 3D and integrated ...
-
Manufactured by Maccura Biotechnology Co., Ltd.based in CHINA
Compatible with i 3000 and i 1000 automatic chemiluminescence analyzer. SARS-CoV-2 IgM, IgG separate detection. Apply to serum and plasma. Pretreatment is not required. Response time is 20 minutes. Open vial expiry date is up to 35 ...
-
Manufactured by Celularity Inc.based in USA
CYNK-CAR candidates are being developed as allogeneic, off-the-shelf approaches through gene modification of human placental hematopoietic stem cell-derived natural killer (NK) cells. Various CAR constructs against hematological and solid tumor targets are under pre-clinical ...
-
Manufactured by Interacousticsbased in DENMARK
The AT235 is an automatic middle ear analyzer ideal for both diagnostic and clinical evaluations. The device also includes pure tone audiometry and the Modified Hughson Westlake audiometry test. +400 to -600 daPa, Direct printing to PC printers, User-defined protocols, HDMI output, Child distraction ...
-
Manufactured by Cognoptix Inc.based in USA
Cognoptix’ SAPPHIRE platform is a compact, easy to use, clinical device for in situ patient examination in the comfort of a doctor’s office. The combined platform is currently available in the United States for investigational use only and is intended for clinical evaluation and measurement of beta-amyloid ...
-
Manufactured by TearLab Corporationbased in USA
of having dry eye disease, in conjunction with other methods of clinical ...
-
Manufactured by SIGA Technologies, Inc.based in USA
and support from BARDA. Formulation prototypes have been generated and will undergo clinical evaluation. ...
-
Manufactured by Genspeed Biotech GmbHbased in AUSTRIA
The FSME IgG xPOC test is an antibody test which provides results in laboratory quality from one drop of blood on-site within 20 minutes. The test detects specific IgG antibodies after vaccination against tick-borne encephalitis (TBE) in laboratory quality. The clinical evaluation study for the GENSPEED FSME IgG xPOC antibody test showed ...
-
Manufactured by MTTS Co., Ltd.based in VIETNAM
, and removable single-infant bassinet. Firefly is designed to treat newborn jaundice in the mother’s room – the best way to support mother-child bonding and breastfeeding. Clinical evaluations indicate that Firefly’s double-sided design reduces total treatment time by 40% on a typical patient compared to single-sided LED phototherapy, ...
-
Manufactured by Vivesto ABbased in SWEDEN
-resistant tumor-initiating cells that are thought to be responsible for disease relapse. In December 2020, top-line results of a Phase I open-label study (NCT02903771), conducted at sites in the USA and Australia, were released. The Phase I study met its primary endpoints, establishing clinical proof of concept, subject to further clinical ...
-
by Vivozon Inc.based in SOUTH KOREA
Single-dose, escalation clinical trial to evaluate the safety and pharmacokinetics of VVZ-149 injection in healthy elderly ...
-
Manufactured by Suzhou PeakSonic Medical Technology Co.,Ltdbased in CHINA
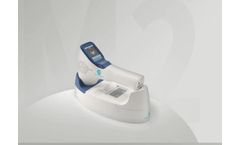
PeakSonic M2 is a handheld portable bladder volume instrument, which can be applied in the department of Surgery, Emergency, ICU, Obstetric, Gynecology, radiotherapy and nursing home. It can provide the basis for clinical catheterization, evaluate the post-void residual after patient urination, and make auxiliary diagnosis on the ...
-
Manufactured by Suzhou PeakSonic Medical Technology Co.,Ltdbased in CHINA
PeakSonic M4 is a handheld portable bladder volume instrument, which can be applied in the department of Surgery, Emergency, ICU, Obstetric, Gynecology, radiotherapy and nursing home. It can provide the basis for clinical catheterization, evaluate the post-void residual after patient urination, and make auxiliary diagnosis on the ...
-
Manufactured by Suzhou PeakSonic Medical Technology Co.,Ltdbased in CHINA
PeakSonic M1 is a handheld portable bladder volume instrument, which can be applied in the department of Surgery, Emergency, ICU, Obstetric, Gynecology, radiotherapy and nursing home. It can provide the basis for clinical catheterization, evaluate the post-void residual after patient urination, and make auxiliary diagnosis on the ...
-
Manufactured by Suzhou PeakSonic Medical Technology Co.,Ltdbased in CHINA
PeakSonic M3 is a handheld portable bladder volume instrument, which can be applied in the department of Surgery, Emergency, ICU, Obstetric, Gynecology, radiotherapy and nursing home. It can provide the basis for clinical catheterization, evaluate the post-void residual after patient urination, and make auxiliary diagnosis on the ...
-
Manufactured by Kiadis Pharma NVbased in NETHERLANDS
K-NK002: A phase 2 clinical study evaluating haplo-identical NK cells to prevent post-transplant relapse in patients with acute myeloid leukemia (AML) and myelodysplastic syndromes (MDS). This study was designed based on promising clinical proof-of-concept data from MD Anderson Cancer Center. The phase 2 trial for K-NK002 will be ...
-
by Vivozon Inc.based in SOUTH KOREA
A randomized, double-blind, placebo-controlled, single and repeated dose, escalation clinical trial to evaluate the safety, tolerability, and pharmacokinetics of VVZ-149 injection in healthy adult ...
-
by Vivozon Inc.based in SOUTH KOREA
A randomized, double-blind, parallel group, placebo-controlled clinical trial to evaluate the analgesic efficacy and safety of VVZ-149 Injection in patients who have pain after artificial hip replacement ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you