Implant Surgical Equipment
-
Manufactured by Glidewell Directbased in USA
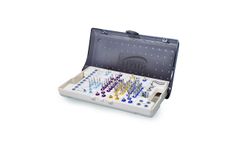
The Hahn Tapered Implant Surgical Kit allows the clinician to easily organize, store, and transport the surgical tooling of the Hahn Tapered Implant System. The autoclavable, color-coded surgical kit clearly maps the drilling protocol for each implant. Drills are arranged from left to right and ...
-
Manufactured by LZQ Tool Co., Ltd.based in CHINA
Temporary abutments are designed to form a gingival sulcus and to preserve papillae during the one stage surgical procedure or after a two stage uncovering procedure. Choose an abutment height that is sufficient to laterally support the interproximal papillae without its being too tall such that it would receive undesirable forces from the ...
-
Manufactured by Glidewell Directbased in USA
The Hahn™ Tapered Implant Guided Surgical Kit allows the clinician to easily organize, store, and transport the surgical tooling of the Hahn Tapered Implant Guided Surgery System. The autoclavable, color-coded surgical kit clearly maps the drilling protocol for each implant. Drills are ...
-
Manufactured by Voyage Medical Solutionsbased in USA
Our VMS team has created partnerships with multiple surgical device companies for our surgeons’ needs. Contact us today for a complete portfolio. If you don’t see a specific implant product you’re looking for, feel free to contact us to see if we can get you the durable medical equipment you ...
-
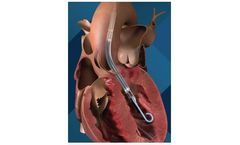
Manufactured by enVVeno Medicalbased in USA
The VenoValve is a first-in-class surgically implanted solution being developed for the treatment of deep venous Chronic Venous Insufficiency (CVI). Implanted into the femoral vein, the VenoValve is designed to act as a one-way valve to help restore proper blood flow up the leg, to return sufficient blood back to the heart. The ...
-
Manufactured by SAEYANG CO.,LTD.based in SOUTH KOREA
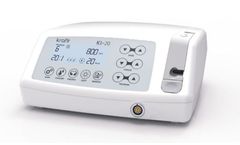
Features: Optic LED Control, Compact Micro motor 40.000 rpm (1:1), 9 memory programs can be stored on single motor, Light & compact optic motor for speedy and accurate ...
-
Manufactured by BioCer Entwicklungs-GmbHbased in GERMANY
TiO2Mesh™ is a surgical mesh implant specially indicated for repair of soft tissue defects of the abdominal wall, where a non-absorbable support material is required. Relevant applications include the repair of inguinal and incisional hernias in all common surgical procedures and even IPOM. Federal law (USA) restricts this device to ...
-
Manufactured by Anatomics Pty Ltd.based in AUSTRALIA
Polymer surgical implants are a permanent, predictable and symmetrical option for facial reconstruction or augmentation. StarPore® implants are an advanced polymer tissue scaffold indicated for the repair or augmentation of contours in the skull and face. Made from clinical proven porous polyethylene (HDPE), animal studies have shown ...
-
Manufactured by Abiomedbased in USA
Impella 5.0 is a temporary, minimally invasive heart pump that provides circulatory support, enabling the heart to rest and recover. Impella LD is a surgically implanted heart pump that provides temporary support to the heart during and after a procedure. Impella 5.0 and Impella LD heart pumps allow patients to walk around while on ...
-
Manufactured by KOS LtDbased in SOUTH KOREA
Titanium Grade 4 / Pure Titanium: Chemical Composition, Specification : ASTM F67. Available Diameter : 3.0~10.00MM. Diameter Tolerance : As per ISO286 , h6. Tensile Strength : As per Customer's requirement ; Levels in 750 Mpa, 850 Mpa, 930 Mpa. Surface : Bright with Centerless Grinding. Packaging : 2.0~4.00 Meter Bar with good straightness for CNC machine. ...
-
Manufactured by RTI Surgicalbased in USA
Non-bone tendons are sterilized through the BioCleanse Tissue Sterilization Process without the use of irradiation. Non-bone tendons used in ACL reconstruction procedures allow for multiple fixation ...
-
Manufactured by RTI Surgicalbased in USA
RTI's Achilles tendons are sterilized through the BioCleanse Tissue Sterilization Process without the use of irradiation. These tendons allow for multiple techniques and fixation options in ligament reconstruction ...
-
Manufactured by RTI Surgicalbased in USA
RTI Surgical offers the only meniscal allograft with both bone and meniscus sterilized through the BioCleanse® Tissue Sterilization Process. The BioCleanse processed meniscus graft provides a sterilized alternative to traditional aseptically processed meniscus allografts and sets the standard for safety and ...
-
Manufactured by RTI Surgicalbased in USA
Matrix HD Allograft Dermis is an acellular human dermis allograft sterilized to a Sterility Assurance Level (SAL) of 10-6 using the Tutoplast Tissue Sterilization Process. This proprietary process retains the three-dimensional intertwined multidirectional fibers and mechanical properties of the native dermis tissue. The Matrix HD graft provides a natural scaffold to support the body's ...
-
Manufactured by RTI Surgicalbased in USA
Conventional allografts are used for various procedures to fill bony voids or gaps in a patient's skeletal system, restore segmental bone loss due to trauma, osteoporosis, removal of tumors or to aid in the surgical correction for a deformity. These grafts provide a scaffold for bone ingrowth to allow for remodeling with the patient’s ...
-
Manufactured by RTI Surgicalbased in USA
RTI Surgical’s fresh-stored osteochondral (OC) allografts enable surgeons to resurface osteochondral defects with mature hyaline cartilage and repair subchondral bone in a single procedure. Fresh grafts are cleansed, processed and preserved to maintain chondrocyte ...
-
Manufactured by RTI Surgicalbased in USA
RTI's bone-tendon-bone (BTB) grafts are sterilized through the BioCleanse Tissue Sterilization Process without the use of irradiation. These grafts allow for multiple techniques and fixation options in ligament reconstruction ...
-
Manufactured by RTI Surgicalbased in USA
Fortiva Porcine Dermis is a non-crosslinked acellular porcine dermal matrix offering a safe and natural biologic option. Fortiva implants are processed through the Tutoplast® Tissue Sterilization Process and terminally sterilized via validated low-dose gamma irradiation to achieve a sterility assurance level (SAL) of 10-6. Fortiva ...
-
Manufactured by RTI Surgicalbased in USA
Cortiva Allograft Dermis is a non-crosslinked acellular dermal matrix offering a safe and natural biologic option and sterilized to a sterility assurance level (SAL) of 10-6 through the Tutoplast® Tissue Sterilization ...
-
Manufactured by RTI Surgicalbased in USA
Whole bones are used as a sterile source of cortical and cancellous bone. These implants may also be used in procedures that restore segmental and critical bone loss in the extremities due to trauma, bone disease, or from the removal of tumors and/or hardware. After being processed through RTI's BioCleanse® Tissue Sterilization ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you